Aurelien Habrioux
University of Poitiers, France
Title: Oxygen evolution reaction at the surface of nickel cobaltites: The impact of surface restructuring phenomena on the activity
Biography
Biography: Aurelien Habrioux
Abstract
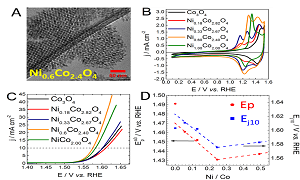
The storage of intermittent renewable energies requires the implementation of efficient energy storage systems. These systems must allow converting renewable energies into sustainable energetic vectors (hydrogen, electron). For this purpose, the oxygen evolution reaction (OER) plays an important role. OER possesses a sluggish kinetics that can be enhanced by using a catalyst exhibiting reliable surface composition and morphostructural properties. To limit the use of scarce noble metals, the synthesis of effective 3d transition metal oxide-based catalysts is of interest. As activity and stability of materials depend on their composition and morphostructural properties, the synthesis of well-defined catalysts is of utmost importance. To this end nanocasting approach constitutes an interesting pathway. In this study, NixCo3-xO4 materials have been synthesized by replicating ordered mesoporous silica templates. Materials were investigated using numerous physico-chemical techniques such as x-ray induced photoelectron spectroscopy (XPS), high resolution transmission electron microscopy, x-ray diffraction and Raman spectroscopy. Evidences from XPS and Raman measurements reveal that the different catalysts surfaces are hydroxylated. A particular attention was paid to restructuring phenomena occurring upon potential cycling and responsible for greatly improving the OER activity. These restructuring phenomena were evidenced using post-mortem Raman spectroscopy and XPS. It was observed that the intrinsic activity of the different restructured catalysts depends on the incorporated nickel amount and correlates with the CoIII/CoIV peak potential. The modulation of CoIII/CoIV peak potential is explained by changes in the chemical environment of surface Co atoms and results in the formation of nickel/cobalt oxy-hydroxide. Nickel indeed modulates the electronic properties of the Co active site and allows improving the OER activity of electrode materials. The catalysts described in this presentation are moreover very efficient since after surface restructuring, the over potential at 10 mA.cm-2 is as low as 310 mV.