Shozo Yanagida
Osaka University, Japan
Title: Verification of photo-splitting of H2O to HOOH and H2 as initial photoproducts
Biography
Biography: Shozo Yanagida
Abstract
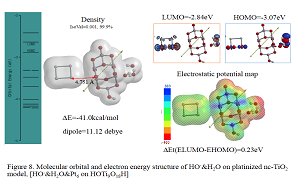
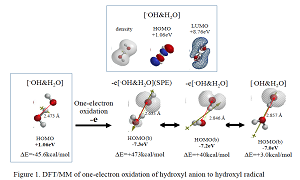
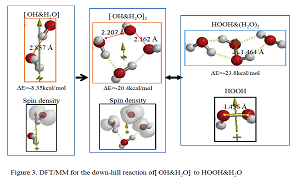
Most electrochemists and biochemists had a mindset that water oxidation yields oxygen molecules. However, Nosaka and his wife reports on generation and detection of reactive oxygen species such as HO. and HOOH in photocatalysis. We verified on the basis of density functional theory-based molecular modeling (DFT/MM) for photoelectrochemical H2O photo-splitting systems that formation of HOOH only under photo-irradiated and highly negative bias conditions. Further literature survey revealed that, in alkali aqueous solutions (pH 8~11.5), Pt-loaded nc-TiO2 catalyzes effective H2O photo splitting to HOOH and H2 as initial products. Figure 8 shows successful DFT/MM for an aggregate induced by van-der-Waals-Coulomb interactions (vdW&Clmb) between HOTi9O18H as a model of nc-TiO2 photocatalyst, HO-&H2O as an alkali water model, and Pt6 as platinum cluster model. Effective photoelectron transfer is verified from [HO-&H2O] to Pt6 for production of H2 on Pt and hydroxyl radical of [HO. & H2O] on nc-TiO2. Figure 1 shows DFT/MM for exothermic one-electron oxidation of alkali water model of hydrated hydroxide anion, [HO- & H2O] to hydroxyl radical of [HO. & H2O]. Figure 2 shows DFT/MM for exothermic vdW&Clmb- induced dimerization of the radical of [HO. & H2O], verifying that oxidation of [HO- & H2O] to HOOH & (H2O)2 via vdW&Clmb dimerization on nc-TiO2. Driving force of photo splitting will be verified as due to highly exothermic electron transfer reaction to Pt6 on nc-TiO2