Day 2 :
Keynote Forum
Pablo Fanjul Bolado
DropSens, Spain
Keynote: ‘In situ’ SERS effect studies with screen-printed electrodes and a compact Raman Spectroelectrochemical Instrument
Time : 09:00-09:25

Biography:
Dr. Pablo Fanjul Bolado, Ph.D in Chemistry by the University of Oviedo (Spain), co-founder and R&D Director of DropSens company has a broad experience dealing with Electroanalytical devices and (bio)sensors. He has focused his research in the design, development and characterization of screen printed electrodes as transducers for biosensing applications in clinical, agri-food or environmental sectors. Working with carbon, gold, platinum and silver electrode surfaces in combination with nanomaterials, he has developed portable analytical solutions to carry out ‘in situ’ measurements in Batch and Flow Injection Analysis systems. Pablo has a great experience in technology transfer from the lab to the market and as entrepreneur, he is encouraged to provide new hyphenated analytical instrumentation to solve analytical problems.
Abstract:
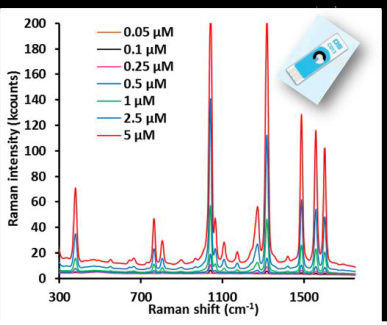
UV-VIS, NIR and Raman Spectroelectrochemistry techniques can be used as ‘in situ’ and real time characterization and quantification techniques. These are powerful techniques since more complete and specific information is obtained with optical and electrochemical signals recorded simultaneously. However, the main current disadvantages are related to complex experimental setup where two analytical instruments (spectrometer and potentiostat) controlled by two different software should be triggered and usually a home-made spectroelectrochemical cell is used [1-3]. The purpose of this study is to introduce a new compact and portable Raman spectroelectrochemical instrument controlled by one software in combination with screen printed electrodes. The new instrument integrates a 785 nm laser, a Raman spectrometer and a potentiostat/galvanostat fully synchronized in the same box. A Raman probe and a Raman spectroelectrochemical cell are used under optimized conditions together with Screen Printed Electrodes (SPEs). These miniaturized strips where the three electrodes of the electrochemical cell are printed together in the same alumina substrate are easy to handle, do not need to be pretreated previously to be used and require a low volume of solution (around 40 μl) [4-5]. Time resolved and quantitative measurements were developed on SPEs using different molecules such as ferricyanide, ruthenium bipyridine, thionine or the active pharmaceutical ingredient called naratriptan. On the other hand, silver, gold and copper based SPEs are shown as cost effective Surface Enhanced Raman Spectroscopy (SERS) substrates. The complete experimental setup is demonstrated to be easy to handle in comparison to the use of conventional electrodes and allows fast, in situ, real time and time resolved Raman Spectroelectrochemical measurements.
Keynote Forum
Sefik Suzer
Keynote: Investigation of Ionic Liquid Interfaces using time- and position-resolved XPS
Time : 09:25-09:50

Biography:
Sefik Suzer has completed his PhD in 1976 from the University of California, Berkeley, USA. After postdoctoral studies at Sydney (Australia) and Freiburg (Germany) Universities, he joined Middle East Technical University in Turkey in 1979 till he moved to Bilkent University in 1992. He has been serving as Editorial Board Memeber of Journal of Electron Spectroscopy, Applied Surface Scince, Spectroscopy and Dynamics, and as an Editor of Surface Scince Reports, and he is s Fellow of the American Vacuum Scince since 2010.
Abstract:
X-ray photoelectron spectroscopy (XPS), a chemical analysis tool, is utilized for investigation of charge screening across metal electrodes fabricated on a porous polymer surface which is infused with an ionic liquid (IL). The IL provides a sheet of conducting layer to the otherwise insulating polymer film, and enables monitoring charging and screening dynamics at the polymer + IL / air interface in a laterally resolved fashion across the electrodes. Time-resolved measurements are also implemented by recording several peaks of the IL, while imposing 10-3 to 10+3 Hz square-wave-pulses (SQW) across the electrodes in a source-drain geometry. Variations in the binding energy of the measured peaks (Au4f, C1s, N1s and F1s) reflects directly the local electrical potential, and allow us visualize screening of the otherwise built-in local voltage drop on and across the electrodes. Accordingly, the device is partitioned into two oppositely polarized regions, each following the polarization of one electrode through the IL medium. Impact of our findings with the presented structure and variants XPS measurements on understanding of various electrochemical concepts will be discussed.
- Corrosion Science and Technology | Physical and Analytical Electrochemistry | Applications of Electrochemistry | Carbon Nanostructures and Devices | Potentiostat Electrochemistry
Location: Berlin, Germany
Session Introduction
Ingo Krossing
Albert-Ludwigs-University, Germany
Title: The Protoelectric Potential Map – Unified Acidity and Reducity Scales and their Experimental Realization

Biography:
Ingo Krossing studied chemistry in Munich (LMU) and finished his Ph.D. thesis 1997 (with Prof. H. Nöth). From 1997 to 1999, he worked as Feodor Lynen postdoc with Prof. J. Passmore at UNB, Canada. In 1999, he started his independent career as a Liebig- and DFG-Heisenberg-Fellow at the Universität Karlsruhe (TH) (mentor: Prof. H. Schnöckel). 2004 he changed as assistant professor to the Ecole Polytechnic Federale de Lausanne (EPFL), before being appointed Chair of Inorganic Chemistry at the Albert-Ludwigs-Universität Freiburg in 2006. His research interests cover ionic systems from reactive cations to ionic liquids, as well as electrochemical energy storage. With an ongoing ERC Advanced Grant he develops absolute acidity and reducity scales.
Abstract:
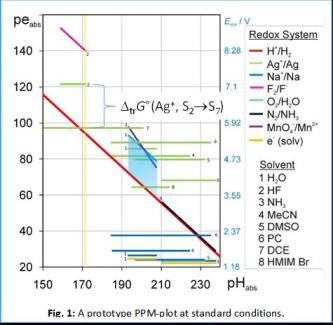
The comparison of acid-base and redox chemistry in their acidity (=> protonation) and reducity (=> electronation) scales is currently limited to measurements within one homogenous medium / solvent. Yet, it is crucial being able to measure chemical potentials of the proton and electron over medium boundaries to overcome this limit. Thermodynamically, the chemical potential differences of proton and electron are straightforward to describe, given the unified reference states ideal proton gas (1 bar) and electron gas (1 bar) we suggested in 2010 (proton gas)[1] and 2014 (electron gas).[2] This unifying concept was developed as part of our in March 2017 ceasing ERC UniChem project. The Protoelectric Potential Map PPM: In analogy to the proto- and electrochemical window of one medium like water, the PPM is a 2D plot of the medium independent pHabs vs. peabs values based on these unifying reference states. An example is shown in Figure 1. Any reaction with transfer of protons and / or electrons in any medium may be placed on the PPM. Given that the respective transfer energies are known (see Figure 1), the thermodynamic relations between them may directly be compared over phase and medium boundaries. This plot reveals at a glance the differences of reducity (i.e. electronation power, synonymous to redox potential) of a redox system (marked by colors) in dependence of the solvent S (marked by digits). For example, the Pearson soft deelectronator Ag+ (green) is up to about 3.5 V or 350 kJ mol–1 more effective in hard solvents like HF (2) or DCE (7) rather than in soft media like NH3 (3) or the Ionic Liquid (IL) HMIM Br (8). In the lecture, we introduce the concept and present reliable ways to measure the necessary transfer quantities over medium boundaries, e.g. between water and acetonitrile or ionic liquids.

Biography:
Huriye Ä°cil was borned in Larnaca, Cyprus, in 1960. She received her Ph.D in Organic Chemistry from Ege University of Turkey, in 1993. In 1993, 1995 and 2002 she was appointed as Assist. Professor, Assoc. Professor and full Professor, respectively at Eastern Mediterranean Univ. (North-Cyprus). She directed many research projects funded by NATO, DFG, DAAD, CNRS, TUBITAK and DPT. She held Visiting Scientists positions at Max Planck Institute (Mulheim), Rochester Univ., and East Anglia Univ. She has 32 published articles in reputed journals. She graduated 33 MSc and 9 PhD students. Her research interests focus on design and synthesis of novel organic materials with remarkable electrical and optical properties.
Abstract:
Development of novel materials with optimal structural, thermal, photochemical and electrochemical properties plays an important role in the researches for a sustainable energy future. Perylene dyes are particularly important compounds due to their remarkable absorption and emission in the spectral region from visible to near infrared (NIR), photochemical and electrochemical properties, and thermal, chemical and photochemical stabilities. They are indeed very promising electron acceptors for efficient organic photovoltaics (OPV). Undoubtedly, photovoltaic applicability of perylene dyes mainly based electrochemical properties. In general, perylene dyes show two different reversible and electrochemically stable reduction peaks in solution state. The first reduction involves the conversion of the neutral compound to the relevant radical anion and the second one accounts for the further reduction to dianion. The separation between the potentials at which the two charge transfers take place allows one to discard the occurrence of any redox reaction between the dianion and the neutral compound. Interestingly the electrochemical characteristics of perylene dyes are completely different in solutions and solid state. They show only one broad and reversible reduction peak in solid state. Further, the analysis of the voltammetric signals suggested that two anodic and cathodic peak systems were actually overlapped. Planarity of the structure, introducing electron donor and acceptor substituents at the imide or bay positions or different intermolecular interactions caused different electrochemical properties. In this study, a detailed comparative study on the electrochemical properties of N-substituted or bay substituted perylene diimides, donor-acceptor-donor structured perylene diimides, self-assembled microstructure perylene diimides and perylene polymers, has been carried out. For comparison, the HOMO-LUMO energy differences for the selected perylene dyes were determined using both electrochemical and optical methods. Notably, the HOMO energy values obtained through the electrochemical and optical band gaps are quite different.

Biography:
Taner Yonar was born in 1974 Turkey. He has a B.Sc. (1996) degree in Environmental Engineering, Uludag University, a M.Sc. (1999) degree in Environmental Science, Uludag University, Institute of Sciences. He has completed his PhD (2005) in Environmental Technology, at Uludag University, Institute of Sciences, where he also worked a research assistant in 1997-2007. He did his post-doctoral research in UK, at Newcastle University, Chemical Engineering and Advanced Materials Department (2011). He is currently working as an Associate Professor at the Environmental Engineering Department of Uludag University.
Abstract:
The application of Sn-Sb-Ni electrodes for the treatment of waste streams is too limited in literature. Sn/SbNi-Ti anodes are tested for electrochemical ozone generation. Sn/Sb/Ni-Ti anodes are promising alloys for ozone production by electrolysis of water because of their stability and over potential for the oxygen evolution reaction. These series of anodes have high electrochemical ozone generation potential at ambient conditions (apprioximately 40% current efficiency). Dairy plant wastewaters are generally high-strength wastes containing soluble, colloidal, and suspended solids at high concentrations, with several sources of chemical and biochemical oxygen demand, but mainly of organic origin. Serious environmental problems can arise if dairy wastewater is not treated properly. According to our knowledge, electrochemical treatment of dairy effluent using Sn/Sb/Ni-Ti anodes is missing in literature. In this study, titanium mesh substrate coated with Sn-Sb-Ni alloy was used as anode immersed in wastewater at room temperature with platinised titanium cathode. Five operational parameters ( initial dye concentration, pH, COD, applied voltage/current and the contact time) were evaluated for the electrochemical degradation of dairy
Toshiaki Kondo
Tokyo Metropolitan University, Japan
Title: Ideally ordered nanohole array obtained by anodizing pretextured metals

Biography:
Toshiaki Kondo works in the field of nano/micro fabrication processing based on electrochemical methods such as anodic oxidation, etching, electroplating and so on. He has proposed several unique fabrication processes of two- and three-dimensionally ordered fine structures and its application to various functional optical devices such as photonic crystals and plasmonic devices. These fabrication processes provide an easy way to obtain various functional devices without applying an electron beam lithography technique.
Abstract:
Anodic porous TiO2 has attracted attention due to its wide range of applications such as photonic crystals, solar cells and so on. Performances of the devices depend on its geometrical structures of a nanohole array. Therefore, for improvement of the performances, control of its geometrical structures is important. Until now, improvement of an arrangement of nanoholes in an anodic porous TiO2 by self-ordering anodization process has been achieved. However, it is still difficult to obtain defect-free nanohole array structures in entire sample area. In this presentation, fabrication processes of an anodic porous TiO2 having ideally ordered nanoholes will be presented. An ideally ordered anodic porous TiO2 was obtained by anodizing a pretextured Ti plate. Through a texturing process, an ordered array of concaves was formed on a surface of Ti. In a subsequent anodization, each shallow concave acts as a starting point for generation of a nanohole at initial stage of the anodization and results in an ideally ordered structure. We demonstrated two pretexturing processes. One is a process to directly texture a surface of Ti. The nanopattern was formed by pressing a metal (Ni) mold onto a surface of Ti. The other is a process to texture a surface of Al layered on a Ti. A two-layered sample composed of an Al top layer and a Ti underlying layer was prepared. An ideally ordered array of concaves was formed on the surface of Al by pretexturing process using a Ni mold. Owing to softness of Al, the nanopattern was easily formed. By anodizing the sample, the arrangement of nanoholes was transferred to the surface of Ti substrate, resulting in the ideally ordered anodic porous TiO2 as shown in Fig. 1. These processes are expected to be applied to fabricate various functional devices, which require ideally ordered nanohole structures in TiO2, such as photonic crystals.

Jonathan Coleman
CRANN and AMBER Research centres, Ireland
Title: All-printed electrochemical thin-film transistors from networks of liquid-exfoliated nanosheets

Biography:
Jonathan Coleman is the Professor of Chemical Physics in the School of Physics and the CRANN and AMBER Research centres, all at Trinity College Dublin. His research involves liquid exfoliation of layered compounds such as graphene, boron nitride and molybdenum disulphide. Exfoliation of these materials gives 2D nanosheets which can easily be processed into thin films or composites from applications from energy storage to sensing to electronics. He has published approximately 250 papers in international journals including Nature and Science, has a h-index of 72 and has been cited ~30000 times. He was recently listed by Thomson Reuters among the world’s top 100 materials scientists of the last decade and was named as the Science Foundation Ireland researcher of the Year in 2011. Prof Coleman has been involved in a number of industry-academic collaborative projects with companies including Hewlett-Packard, Intel, SAB Miller, Nokia-Bell Labs and Thomas Swan..
Abstract:
With the advent of the Internet of Things, the development of printed electronics (PE) is becoming increasingly important, with much research focusing on developing new materials. A number of material sets have been studied, including organics, inorganic nanoparticles and nanotube/nanowire networks. High operating voltages (up to 50V), low mobility (<10 cm2/Vs) and poor current injection are still challenges for organic thin film transistors (OTFTs). Networks of inorganic nanoparticles or nanotubes have demonstrated mobilities and on:off ratios of >10 cm2/Vs and >106 respectively, but may face problems with scalability and integration. These problems have led a number of researchers in the field of 2D materials to attempt to produce printed transistors where the channel material is a network of semiconducting nanosheets. Because of the relatively high mobility of 2D materials, such a network might display mobilities which are competitive or even superior to those achievable with printed organics. In addition, one could envisage all-printed transistors consisting of interconnected networks of semiconducting, conducting and insulating 2D nanosheets. However, switchable nanosheet networks have not been demonstrated. Here, using electrolytic-gating, we demonstrate all-printed, vertically-stacked electrochemical transistors with graphene source, drain and gate electrodes, a transition metal dichalcogenide channel and a BN separator, all formed from nanosheet networks. The BN network contains an ionic liquid within its porous interior that allows electrolytic gating in a solid-like structure. Nanosheet network channels display on-off ratios of up to 600, transconductances exceeding 5 mS and mobilities of >0.1 centimeters squared per volt per second. The on-currents scaled with network thickness and volumetric capacitance as well as the network mobility. In contrast to other devices with comparable mobility, large capacitances, while hindering switching speeds, allow these devices to carry higher currents at relatively low drive voltages.to become trauma- informed that would help this recognition.

Vardan Galstyan
University of Brescia, Italy
Title: Electrochemical surface modification of metal oxides for security applications and healthcare

Biography:
Vardan Galstyan is now researcher at the Sensor Lab, Department of Information Engineering, University of Brescia (Italy). His activity is dedicated to the developments of the synthesis methods using electrochemical approaches for the preparation of nanomaterials and nanocomposites (mainly metal oxides and graphene based materials). He has worked on characterization of the obtained structures for the applications in gas- and biosensors, energy storages, photocatalysis and biomedicine. V. Galstyan is serving as peer reviewer for the different international journals including Journal of Materials Chemistry A, Journal of Materials Chemistry C, ChemComm, ACS Applied Materials & Interfaces, Nanoscale, Physical Chemistry Chemical Physics, Sensors & Actuators: B. Chemical, CrystEngComm, etc.
Abstract:
Statement of the Problem: Chemical sensors have received great attention for the fabrication of small-size and mobile gas sensing devices for the environmental monitoring and clinical diagnostic.1,2 One-dimensional metal oxide nanomaterials are very promising structures for the application in chemical gas sensing.3,4 However, the sensing performance of these materials needs to be improved for the manufacturing of sensing systems with the high sensitivity, selectivity and stability.5 Herein, we report the synthesis of highly ordered titania nanotubes and the modification of their structure and surface for the chemical sensing applications. Methodology & Theoretical Orientation: Well-ordered titania nanotube arrays were prepared by electrochemical anodization. The anodization process was performed in the electrochemical cell with the two-electrode system. Preparation of titania nanotubes by means of electrochemical anodization is anodic formation of nanotubes by oxidation and etching of metallic titanium films. Pt foil was used as a counter electrode. The anodization was carried out by potentiostatic mode at room temperature. This method allows direct growth of the titania nanotubes on different type of substrates and the modification of their surface structure at room temperature. The morphological, structural and elemental analysis of the obtained samples were performed. The sensing properties of the materials were tested towards different, explosive and toxic gases. Findings: Pure and doped nanotubes have been obtained by the modification of anodization parameters. Our studies have shown that the engineering of the band gap and the functionalization of the surface structure of titania enhanced its gas sensing performance. Conclusion & Significance: Investigations have shown that the response and the selectivity of titania nanotubes are improving depending on the modification of their composition and their functionalization. Meanwhile, the developed method is promising for the fabrication of high performance and small size chemical gas sensors.

Biography:
Benoît Ter-Ovanessian has been named associate professor in the Material Science and Engineering Department at INSA de Lyon in France in 2013. He has joined the Corrosion Science and Surface Engineering group of the MATEIS laboratory and integrated the research project of the CNRS International Associated Laboratory, ELyT Lab, which promotes collaborations between Lyon University (Fr) and Tohoku University (Jp). His research activities are mainly focused on the understanding of the passivation features, the interactions between mechanical loading and interfacial reactivity; and consequently the managing of the reliability of metallic components.
Abstract:
Semi-conductive properties of passive film represent discriminant parameters in many applications in photo-electrochemistry such as photoanodes or photoelectrochemical cells, or in electrochemistry such as sensors, bio-electrochemical cells and corrosion. For this reason, the accurate determination of these properties is of great interest. Considering passive films as thin highly disordered semiconductor, the electronic properties of passive films are generally investigated by photo-electrochemistry or differential capacitance measurement of the Mott-Schottky (MS) theory. In the MS approach, electrochemical impedance spectroscopy (EIS) is a widely used technique to determine the capacitance of the passive film. However, different limitations are known to alter the final results. For example, the electronic properties obtained by conventional MS experiments are known to be frequency dependent. Furthermore, as Constant-Phase-Element (CPE) behavior is commonly observed on the impedance diagrams recorded with passive materials, the method to extract the effective capacitance from the high frequency time-constant dispersions has to be carefully chosen.
In the present work, the recent advances in experimental procedure and interpretations of EIS spectra during MS experiments, which allow to accurately determine the electronics properties of passive film, are described for passive films grown on pure Chromium and Nickel-Chromium alloys. The relevance of multi-frequency MS experiments that limits the frequency dependency of the capacitance measurements is firstly discussed. Secondly, the pertinence of the power-law model or the Cole-Cole representation of the complex capacitance to assess the passive film capacitance from EIS diagrams during MS experiments is then debated for different systems.

Biography:
Dong Junhua is a professor working in Institute of Metal Research (IMR), Chinese Academy of Sciences (CAS). He has his expertise in electrochemical corrosion principle of metals in study and teaching. His primary research aim is to solve various industrial and natural environmental corrosion problems. His research interests has been focused on: the evaluation of atmospheric corrosion evolution of low alloy steels; the study of developing cost effective weathering steel and marine corrosion resistant steel; the design of waste containers for the underground geological disposal of high level nuclear wastes in China; the study of marine corrosion in tidal zone and deep sea; the evaluation of concrete corrosion; the study of magnesium corrosion and protection by alloying and coating technique; the evaluation of corrosion inhibitors; electrochemical corrosion monitoring for various corrosion environments. He has taught the course of electrochemical corrosion principle of metals to the graduate students for 14 years in his organization.
Abstract:
Statement of the Problem: Low carbon steel with the microstructures of ferrite and pearlite phases is the most widely used engineering structure material, in which pearlite with a typical lamellar structure consisting of alternating layers of ferrite and cementite (Fe3C) plays an important role in the mechanical properties of steel. However, as the electrically contact of Fe3C as a cathode site and ferrite site as anode in electrolyte can cause the galvanic corrosion, the corrosion resistance of steels will be rapidly deteriorated. The purpose of this study is to describe the sustained effect of the remaining cementite on enhancing the corrosion deterioration and the effect of several added alloy elements on retarding the corrosion of low carbon steel in an acid solution. Methodology & Theoretical Orientation: Immersion test, X-ray diffraction, micromorphology observation, potentio-dynamic polarization, open circuit potential monitoring and electrochemical impedance spectroscopy was utilized in this study. Findings: The results show that, with extending the immersion time, the preferential dissolution of the exposed ferrite resulted in a lot of cementite accumulation on the steel surface, which enhanced the galvanic effect and hence accelerated the corrosion. The promoted anodic dissolution of ferrite phases was controlled by charge transfer process, while the hydrogen evolution reduction occurred on cementite was controlled by the diffusion process. For 16MnCu steel, elemental Cu precipitated as the nano-sized particles on the steel surface, which improved the corrosion resistance of the steel by weakening the galvanic effect between the ferrite and cementite sites. Moreover, trace of alloy elements of Sn or Sn-Mo could improve the corrosion resistance of AH32 steel, which is also ascribed to that Sn or Sn-Mo could retard the galvanic corrosion between the ferrite and cementite phases. Sn and Mo should exist as the metallic state and were uniformly distributed on the steel surface.
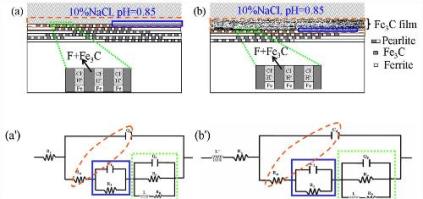
Nagahiro Hoshi
Chiba University, Japan
Title: Structural effects on the oxygen reduction reaction on single crystal electrodes Pt modified with amines

Biography:
Nagahiro Hoshi is a professor of Department of Applied Chemistry and Bio-technology, Graduate School of Engineering, Chiba University, Japan. He has his expertise in surface electrochemistry. The main target of the study is the elucidation of surface structures of electrocatalysts that have high activity and selectivity.in electrochemical reactions He can prepare well-defined single crystal electrodes of Pt, Pd, Ir, Rh, Au, Cu, Pt alloys and TiO2 etc. in his laboratory. He can also synthesize shape controlled nanoparticles of Pt and Pt alloys. The real surface structures of the well-defined electrodes are determined using surface X-ray diffraction, vibrational spectroscopy and probe microscopy. His present research projects are the reduction of Pt loading in electrocatalysts of fuel cells, enhancement of conversion efficiency of dye sensitized solar cell.
Abstract:
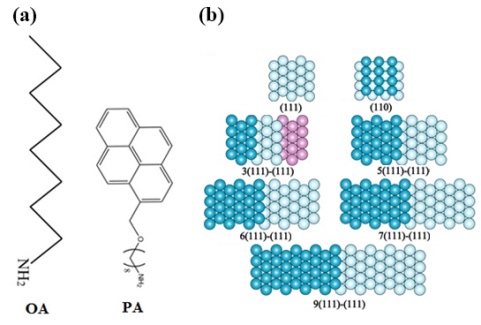
Enhancement of the activity for the oxygen reduction reaction (ORR) is important for the development of fuel cells. Structural effects on the ORR have been studied using single crystal electrodes of Pt1-5, Pd6, Pt3Ni7 and Pt3Co8. Recently, Miyake et al, reported that Pt nanoparticles modified with amines such as OA and PA (Fig. 1(a)) give higher activity for the ORR and durability than bare Pt nanoparticles. We have studied structural effects on the enhancement of the ORR by the amines on n(111)-(111) surfaces of Pt that have the highest activity for the ORR.4 In the notation n(hkl)-(mno), n, (hkl) and (mno) shows number of terrace atomic rows, structure of terrace and step, respectively. Hard sphere models of the surfaces examined are shown in Fig. 1(b). The ORR voltammograms were measured in 0.1 M HClO4 saturated with O2 using rotating disc electrode (RDE) at room temperature. The activity for the ORR was estimated using specific activity at 0.90 V(RHE) jk. OA/PA = 9/1 (molecular ratio) was adsorbed on a single crystal surface of Pt. The peaks of hydrogen adsorption/desorption and oxide formation in voltammograms shrink after the modification of OA/PA in Ar saturated solution. The value of jk of Pt(111) modified with OA/PA was 2.5 times as high as that of bare Pt(111). On Pt(331) = 3(111)-(111) that has the highest activity for the ORR in n(111)-(111) surfaces, however, the value of jk after OA/PA modification is lower than that of the bare surface. Detailed study on the structural effects shows that OA/PA enhances the ORR activity on the surfaces with terrace atomic rows n ≥ 7. On the surfaces with n ≤ 6, OA/PA deactivates the ORR. We have also studied the OA/PA effect on Pt(100) of which surface is composed of flat terrace with 4-fold symmetry. The ORR activity on Pt(100) is deactivated after the OA/PA modification. OA/PA enhances the ORR activity of Pt electrodes with more than 7 atomic rows of (111) terrace, but deactivating the ORR on narrow (111) surfaces with terrace atomic rows less than 6 and (100) structure. The reason of the enhancement of the ORR activity by OA/PA has not been elucidated. DFT calculation attributed the higher activity on n(111)-(111) surfaces of Pt compared with Pt(111) to the destruction of well-ordered structure of adsorbed water on (111) terrace.9 OA/PA may also change the adsorbed water structure on wide (111) terrace. Structure of adsorbed water needs to be elucidated using vibrational spectroscopy.
References
- N. M. Marković, R. R. Adzić, B. D. Cahan, E. B. Yeager, Structural effects in electrocatalysis: oxygen reduction on platinum low index single-crystal surfaces in perchloric acid solutions, J. Electroanal. Chem. 377, 249 (1994).
- M. D. Macia, J. M. Campina, E. Herrero, J. M. Feliu, on the kinetics of oxygen reduction on platinum stepped surfaces in acidic media, J. Electroanal. Chem. 564, 141 (2004).
- A. Hitotsuyanagi, M. Nakamura, N. Hoshi, Structural effects on the activity for the oxygen reduction reaction on n(111)-(100) series of Pt: correlation with the oxide film formation, Electrochim. Acta. 82, 512 (2012).
- N. Hoshi, M. Nakamura, A. Hitotsuyanagi, Active sites for the oxygen reduction reaction on the high index planes of Pt, Electrochim. Acta. 112, 899 (2013).
- F. Sugimura, M. Nakamura, N. Hoshi, The oxygen reduction reaction on kinked-stepped surfaces of Pt, Electrocatalysis, 8, 46-50 (2017).
- S. Kondo, M. Nakamura, N. Maki, N. Hoshi, Active sites for the oxygen reduction reaction on the low and high index planes of palladium, J. Phys. Chem. C, 113, 12625-12628 (2009).
- T. Rurigaki, A. Hitotsuyanagi, M. Nakamura, N. Sakai, N. Hoshi, Structural effects on the oxygen reduction reaction on the high index planes of Pt3Ni: n(111)-(111) and n(111)-(100) surfaces, J. Electroanal. Chem., 716, 58–62 (2014).
- Y. Takesue, M. Nakamura, N. Hoshi, Structural Effects on the Oxygen reduction reaction on the high index planes of Pt3Co, Phys. Chem. Chem. Phys., 16, 13774-13779 (2014).
- R. Jinnouchi,K. Kodama, Y. Moromoto, DFT calculations on H, OH and O adsorbate formations on Pt(111) and Pt(332) electrodes J. Electroanal. Chem. 716, 31 (2014).
Sandra Luber
University of Zurich, Switzerland
Title: Exploring ionic liquids and solar water splitting catalysis via high-performance ab initio molecular dynamics

Biography:
Sandra Luber received her MSc and PhD degree from ETH Zurich in 2007 and 2009, respectively. After postdoctoral studies at Biozentrum of the University of Basel (2010) and Yale University (2010-2011), she joined BASF SE in 2012 followed by a position as project group leader at University of Zurich. Currently, she is SNSF professor at the University of Zurich. Awards include the ETH medal for an outstanding PhD thesis, the IBM Research Prize for Computer Modelling and Simulations in Chemistry, Biology, and Materials, and the Clara Immerwahr Award 2017.
Abstract:
Ionic liquids receive more and more attention due to their potential applications as, for instance, electrolytes and green solvents. Being composed of charged entities, they exhibit favorable properties of high interest for use in laboratories and industry. In order to study their complex behavior, dynamic ab initio calculations are indispensable. The same is true for the understanding of water splitting catalysts in solution. Detailed analysis of their catalytic functioning and the factors determining their efficiency are a prerequisite for the design of more efficient catalysts. We present our recent research for the in-depth study of ionic liquids and water splitting catalysis using forefront computational methods such as high-performance ab initio molecular dynamics.
Chunwen Sun
Beijing Institute of Nanoenergy and Nanosystems, CAS, China
Title: Recent progress in bifunctional catalysts for rechargeable Li-air batteries

Biography:
Chunwen Sun is a Professor at Beijing Institute of Nanoenergy and Nanosystems (BINN), Chinese Academy of Sciences (CAS). He received his PhD degree in Condensed Matter Physics from the Institute of Physics (IOP), CAS in 2006. After graduation, he worked as a Post-doctoral Research Fellow at Technische Universität München (TUM), Germany, then joined at Institute for Fuel Cell Innovation, National Research Council Canada (NRC) as a Research Associate Officer; he has also worked as a Post-doctoral Research Fellow with Professor John B Goodenough at the University of Texas at Austin in 2010~2011. He has been a Full Professor and Group Leader of Energy Storage Materials and Devices in BINN since 2015. His current research interests include energy storage and conversion, e.g., lithium/sodium-ion batteries, metal-air batteries, all-solid-state batteries, fuel cells and self-power systems. He has published more than 80 peer-reviewed papers with a citation >3200 times, edited 4 book chapters and filed 13 Chinese patents. He has also received a number of awards, including the Outstanding Overseas Talents by the Institute of Physics CAS (2011), the Second Prize of Military Progress Prize in Science and Technology Advancement in China (2013) and the International Association of Advanced Materials (IAAM) Scientist Medal (2017).
Abstract:
Among various metal/air batteries, lithium-air batteries possess the highest theoretical gravimetric energy density. However, for the rechargeable Li-air battery, besides the challenges of cost, another issue that has to be addressed in the current technology is the limitations of oxygen reduction reaction (ORR) during discharging process and oxygen evolution reaction (OER) during charging process. The sluggish kinetics of the ORR and OER in Li-air batteries is ascribed to the low efficiency of catalysts. Therefore, the design of a low-cost and stable bifunctional electrocatalyst is a major challenge to the construction of efficient Li-air batteries. Several nanocomposite bifunctional catalysts with low cost have been developed in our lab and studied for lithium-air battery applications. They show enhanced catalytic activity and better long-term durability than the commercial Pt/C catalyst.
Atsushi Kitada
Kyoto University, Japan
Title: Electrochemical properties of AlCl3/glyme baths for room temperature aluminum electrodeposition

Biography:
Atsushi Kitada is an Assistant Professor in the Department of Materials Science and Engineering at Kyoto University, Japan, since 2012. His main research topics are focused on synthesis and preparation of liquid and solid materials with electrochemical, electronic, and magnetic properties. He focuses on liquid state properties and electrochemical characterization of less volatile non-aqueous electrolyte solutions, which may be used as next-generation batteries and electroplating. He is also interested in solid state properties e.g. magnetic and electronic properties of oxides. He has done his PhD (Dr. Eng.) from the Department of Energy and Hydrocarbon Chemistry, Kyoto University.
Abstract:
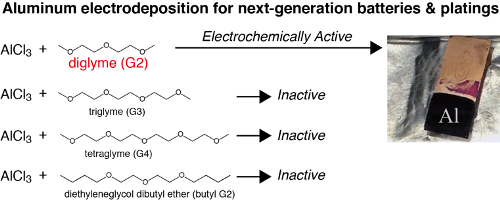
Aluminum (Al) metal, known for its low weight and corrosion resistance, finds its applications in structural materials. Smelting of aluminum has been industrialized as the Hall-Héroult process. Electrowinning of aluminum from alumina-dissolved cryolite molten salts is performed at 1000ºC. Instead of the high temperature process, medium and low temperature electrodeposition is fascinating as that can be hired as a new aluminum coating process. Several kinds of medium-low temperature baths for aluminum electroplating are reported using organic solvents (ethers, aromatic hydrocarbons and sulfones) and ionic liquids. Al electroplating is employed for the enhancement of corrosion resistance of magnesium alloys and steel. Additionally, the redox potential of aluminum is low and the theoretical capacity is high. Hence aluminum electroplating baths are captivating as a negative electrode material for the aluminum ion batteries. At electrodeposition temperatures, the usual organic solvents are highly volatile. Since ionic liquids are thermally and chemically stable and also less volatile at room temperature, they are adorable for electroplating baths. On comparing ionic liquids with organic solvents, however, cost of the chemicals required for the bath preparation is high for ionic liquids. Finding a cost effective and safe substitute for aluminum electroplating baths is important. Currently glymes, i.e. glycol ethers, have found to be relatively safe solvents for lithium and magnesium ion batteries, which have boiling points above 150ºC and relatively low volatilities at room temperature. The work reports of room temperature aluminum electrodeposition using aluminum chloride (AlCl3) and diglyme (G2) is the first report for aluminum electroplating glyme baths. We also studied the electrochemical properties of AlCl3/glyme solutions using four kinds of glymes. By surprise, electrodeposition was only successful from G2 solutions: only G2 can form electrochemically active Al-Cl-glyme complex cations, which can then undergo easy desolvation of the glymes and subsequent reduction of Al3+.
Nisrine Benzbiria
University of Chouaib Doukkali, Morocco
Title: Determination of the kinetic parameters of oxygen reduction on copper using a rotating disk electrode in neutral medium

Biography:
Nisrine BENZBIRIA is a PhD student currently attending Faculty of Sciences of El Jadida at Chouaib Doukkali University, with a joint supervision in Faculty of Sciences of Casablanca at Hassan II University. Her current research project takes part in the study of the kinetic parameters of oxygen reduction reaction on copper and aluminium, which are widely used in aeronautical industry. Prior to her doctoral studies, she received her engineering degree in materials from Cadi Ayyad University in 2012. Then, she worked as a head of slip casting Department in Casablanca.
Abstract:
Regarding their excellent mechanical properties associated to a corrosion resistance quite satisfactory, the passivated metals occupy a prominent place in several industries. However, these materials undergo corrosion in some media especially those containing chloride. Consequently, to understand the phenomena taking place into their surface, some electrochemical investigations should be done. The majority of the previous studies concerned only anodic dissolution of the metal while the cathodic reaction, which is least studied, can provide relevant information. Oxygen reduction assumes importance in corrosion processes since in neutral solutions and in the absence of any other depolarizer the corrosion rate is controlled by the kinetics of oxygen reduction reaction. Our interest is to outline the significant role of oxygen reaction reduction (ORR) on copper. For this purpose, we have used the rotating disc electrode (RDE) to obtain cathodic polarization curves in neutral Na2SO4 solution containing different O2 concentrations. Three types of copper surfaces were investigated: pre-reduced electrodes, copper pre-oxidized to Cu/Cu2O and pre-oxidized to the duplex Cu/Cu2O/CuO layer. This will allow us to elucidate the influence of film and dissolved oxygen concentration on the kinetics of the cathodic reaction. Parameters, such as reaction order, kinetic current, Tafel slopes as well as the number of electrons transferred are determined.
Siham Echihi
Chouaib Doukkali University, Morocco
Title: Inhibition of copper corrosion in 0.1m NaCl solution by 1.3.4 thiadiazole 2.5 dithiol (dmtd): Experimental and theoretical study

Biography:
SIHAM ECHIHI is a PhD student currently attending Faculty of Sciences of El Jadida at Chouaib Doukkali University, with a joint supervision in Faculty of Sciences of Rabat at Mohamed V University, her research interests are Corrosion inhibition of aluminium alloys rich with copper for aeronautical applications, she received the Bachelor and Master degrees in Chemistry Science from the Faculty of Sciences at University Mohamed V of Rabat in 2008 and 2010, respectively.
Abstract:
Corrosion of copper and its inhibition by 1.3.4 thiadiazole 2.5 dithiol (DMTD) have been investigated in 0.1M NaCl solution using potentiodynamic polarization (PDP), electrochemical impedance spectroscopy (EIS), scanning electron microscopy (SEM) along with energy dispersive (EDX), atomic absorption spectroscopy (AAS) and ion chromatography (IC), quantum chemical calculations and the molecular dynamics (MD) simulation. Relationships between inhibition efficiency and molecular orbital were also evaluated. PDP measurements showed that the presence of DMTD significantly decreases anodic, cathodic, and corrosion currents in the studied solution with more pronounced effect on anodic domain and the inhibition efficiency decreases with increasing the temperature. EIS measurements indicate that the values of polarization resistance and inhibition efficiency tend to increase by increasing the immersion time inhibitor. SEM/EDX measurements revealed that DMTD is adsorbed on the copper surface at open-circuit potentials, preventing copper from being corroded by forming a protective layer on its surface. IC and AAS analysis results showed that the concentrations of chloride ions and copper(II) ions in 0.1M NaCl increased and decreased, respectively, after the corrosion process, when 10-2 M of DMTD is present in the solution. Quantum chemical calculations and the molecular dynamics (MD) simulation show that DMTD has relatively small ΔE between HOMO and LUMO and large negative charge in its three sulfur atoms, which facilitates the adsorption of DMTD on the copper surface. Therefore, the studied molecule is likely to adsorb on the copper surface in aqueous phase to form stable adsorption layers and protect copper from corrosion.
Ismail Abidat
University of Poitiers, France
Title: Highly Active and Stable Heteroatoms-Doped Supported Spinel Nanoparticles as Bi-functional Oxygen Electrocatalysts

Biography:
Ismail Abidat has a Master degree in material sciences. He is presently in his third year of PhD in electrocatalysis under the supervision of Dr. A. Habrioux, Dr. C. Morais and Dr. T. Napporn, at the Institute of Chemistry of Poitiers, IC2MP in the university of Poitiers. (France). His research focuses on innovative nanomaterials with advanced properties, which are useful in energy conversion and storage systems. He is developing various hybrid materials of mesoporous and spinel metal oxides nanoparticles with heteroatoms doped graphene. These stable nanocomposites are reversible electrodes for both oxygen reduction reaction (ORR) and oxygen evolution reaction (OER). They are very good candidates for air electrodes in rechargeable metal-air batteries.
Abstract:
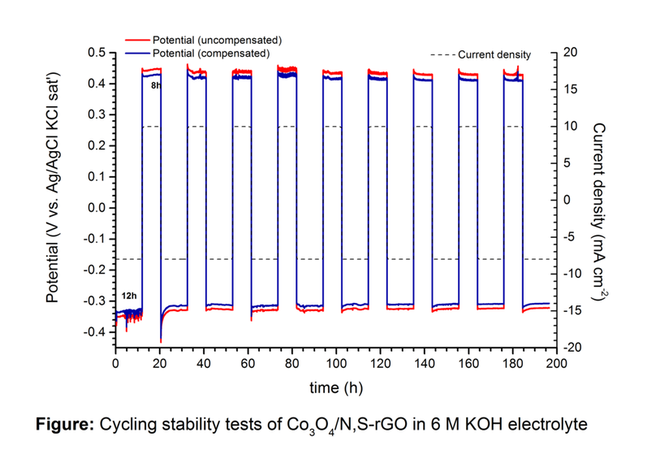
Rechargeable alkali Metal-air batteries are one of the most promising energy storage systems due to its extremely high theoretical energy density. However, its development requires the design of effective and robust bi-functional electrocatalysts that act as air electrode for oxygen evolution (OER) and oxygen reduction (ORR) reactions. Especially, reduction of overpotentials, promotion of reaction kinetics and long term reliability under repeated OER and ORR cycles are some of the main features of an optimal high-performance electrocatalyst. In this context, composite electrode materials have been recently synthesized by coupling heteroatom-doped graphene like materials with spinel-type transition metal oxides. This combination leads to the formation of oxide-carbon heterointerfaces able to enhance the electrocatalytic activity of materials towards OER and ORR as well as their stability. Therefore, in the present work, we have synthesized nanocomposite catalysts made of Co3O4 nanoparticles deposited onto various nitrogen-doped as well as onto nitrogen and sulfur dual-doped graphene oxide materials as non-noble bifunctional electrocatalyst using an original microwave heating method. The heteroatoms-doped graphene materials were prepared using hydrothermal and thermal methods in order to control ratio and nature of Nitrogen functionalities. The crystallinity of the nanocomposites was confirmed by X-ray diffraction (XRD). Transmission electron microscopy (TEM) allowed investigating morphological characteristics. Rotating Ring disk (RRDE) measurements revealed that the value of the reversibility criterion could be tuned depending on the chemical nature of the heteroatom-doped graphene-like material and on the morphostructural properties of the different catalysts. We also investigated the stability of the Co3O4/N,S-RGO in harsh conditions of repeated OER and OER cycles. In 6 M KOH at 10 mA cm-2 (12 h per cycle) in OER and 8 mA cm-2 (8 h per cycle) in ORR, no loss in activity was detected after 200 h (Figure 1). The obtained results show that the synthesized nanocomposites are promising air electrode candidates for metal-air batteries.
KristÃna Cinkova
Slovak University of Technology, Slovakia
Title: Microdisc versus Microcylinder Electrodes: Detection of Solution Phase Trace Oxygen

Biography:
Kristína Cinková has graduated in Analytical Chemistry in 2014 and is a PhD student at the Faculty of Chemical and Food Technology, Slovak University of Technology in Bratislava, Slovakia. She is interested in characterisation and utilisation of novel electrode materials in determination of biologically active compounds in various matrices. Her contribution to carbon microdisc electrodes was done within the scholarship programme at the University of Oxford. At her young age, she is author and co-author of 14 papers.
Abstract:
The specific apparent capacitance (Farads per unit geometric area) of commercial carbon microdisc electrodes and in-house fabricated carbon microcylinders is studied through the use of cyclic voltammetry. For accurate electroanalytical measurements, three important considerations need to be taken into account. A reduced geometric area does not necessarily lead to practically low capacitances due to factors such as surface roughness, poor sealing and potential microfractures leading to a significant variation in experimentally observed capacitance. Comparing the specific apparent capacitances of working electrodes, the capacitance of fabricated carbon microcylinder electrode was found to be lower by a factor of 35 despite the fact that both type of electrodes were made of nominally the same material. As a consequence of the reduced capacitance, carbon microcylinder electrodes were experimentally proven to be superior in terms of analytical sensitivity over commercial carbon microdisc electrodes. We further demonstrate practically how the electroanalytical limit of the detection of solution phase trace oxygen is significantly improved using such microcylinder electrodes. Finally we show that after the degassing of a solution by nitrogen purging even in the most commonly encountered ‘best’ case scenarios approximately tens of mM of oxygen remain present in the electrochemical cell, unobservable by the commercial microdisc electrodes, but detectable with microcylinder electrodes, testifying to their analytical superiority. This work was supported by the Grant Agency of the Slovak Republic (Grant No. 1/0489/16) and Grant scheme for Support of Excellent Teams of Young Researchers.
Enrico Pizzutilo
Max-Planck Institute for Iron Research, Germany
Title: On-site production of hydrogen peroxide in electrocatalysis and in heterogeneous catalysis

Biography:
Enrico Pizzutilo is a PhD student in the Electrocatalysis Group of Prof. Dr. Karl Mayrhofer at the Max-Planck Institut for Iron Research. His expertise and interest are electrocatalyst for fuel cell and electrolyzer application as key technologies for solving the current energy storage problem. The current work has been elaborated in collaboration with the group of Graham Hutchings at the Cardiff Catalysis Institute, where he spent part of his PhD. The two groups developed a new electrocatalytic technique to help describing the heterogeneous catalytic processes when electron transfer is involved.
Abstract:
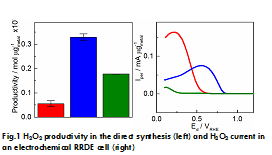
Simon Geiger
Maxplank Institute for Iron Research, Germany
Title: Stability challenges of iridium-based oxides towards acidic water splitting – Structure matters!

Biography:
Simon Geiger has his expertise in stability evaluation of electrocatalysts for the oxygen evolution and oxygen reduction reaction. He studied chemical engineering and chemistry at the applied university of Mannheim and university of Stuttgart, before he joined the electrocatalysis group of Karl J. J. Mayrhofer at the Max-Planck-Institut für Eisenforschung in Düsseldorf, where he is currently working on his PhD.
Abstract:
Reduction of noble metal loading and increase of specific activity are omnipresent challenges for oxygen evolution catalysts in proton exchange membrane (PEM) water electrolysis. The latter are often tackled by using iridium oxides with amorphous1or perovskite2,3structure with less focus on their stability during operation. In this presentation degradation processes of various iridium-based perovskites in relation to amorphous and crystalline iridium oxide are explored: Leaching of the non-noble elements in perovskites will lead to formation of amorphous iridium oxide, which is very active towards oxygen evolution but does not fulfil stability requirements4. Crystalline IrO2, on the other hand, resists the harsh operational OER conditions to a great extent, however, higher potentials have to be applied to reach the same current density. Combination of data on activity, dissolution, and structure are summarized in a conclusive dissolution pathway which is correlated with OER mechanisms. The instability of amorphous structures is explained by participation of activated oxygen atoms5, generating short-lived vacancies that favor dissolution. These insights are considered to guide further research which should be devoted to increasing utilization of pure crystalline iridium oxide, as it is the only known structure that guarantees lifetimes ≥10 years in acidic conditions. In case amorphous iridium oxides are used, solutions for stabilization are needed.
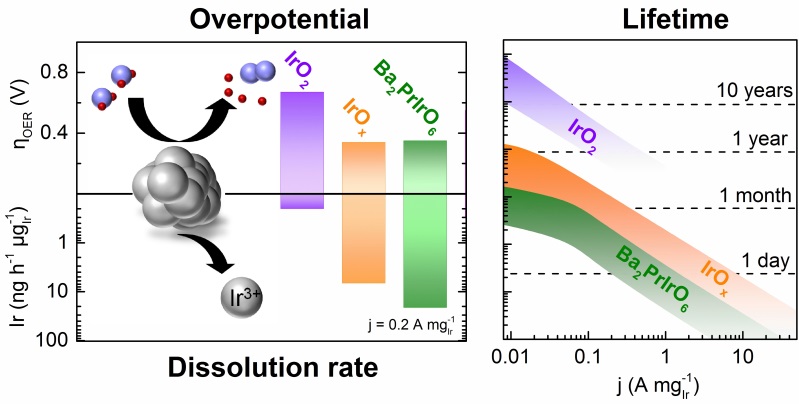

Biography:
Andrés G. Muñoz is a senior scientist passionate about fundamental questions of electroplating of semiconductors and metals. He has gained his expertise by working with renowned scientists from the German electrochemistry school at the Heinrich-Heine University of Düsseldorf and the Research Centre Jülich. After being distinguished with an Alexander von Humboldt fellowship, he succeeded in combining his wide background in electrochemistry with surface science on joining the Institute for Solar Fuels and Energy Storage Materials at the Helmholtz-Center in Berlin. He resolved fundamental questions of interfacial photoelectrochemical processes in solar energy conversion systems. Presently, his is also involved in projects concerning the application of electrodeposition to improve the corrosion
Abstract:
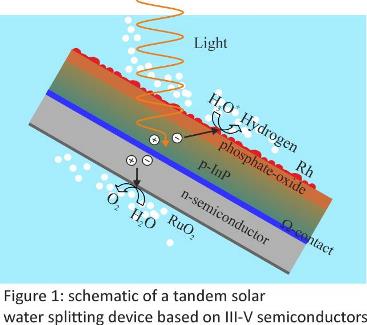
Solar water splitting tandem devices are a promising route for building a carbon-neutral energy infrastructure based on hydrogen as a fuel. The development horizon suggests the use of technologically advanced semiconductor materials, mainly III-V ones, due to its fast technical realization. The coupling of high-efficiency photovoltaics with electrochemical water splitting processes (two in one systems) requires from smart conditioning methods to turn semiconductor surfaces electrochemical active (figure 1). Particularly, the decoration of with noble metals, such as Pt and Rh is one of the most pursued approaches in the design of photocathodes to be integrated in tandem-type devices. Electroplating is preferred method due to its simplicity, its low cost and its power for accessing hidden places in complicated geometrical shapes (structured surfaces with enhanced photonic properties). The optical and electrocatalytic performance of deposited metal films can be enhanced by achieving a particular size and distribution of their nano-dimensioned grains. The chemical composition of the electrolytic bath, the applied potential program and illumination intensity constitute the main tools to do this. Electroplating of noble metals, however, triggers interfacial reactions generating chemical and electronic surface transformations. This is due to the complex multi-electron transfer involved in the metal phase formation and the particular high thermodynamic redox potential. Therefore, the selection of a galvanic method must balance coupled interfacial processes with desired properties of the electrocatalytic film. The complex picture of electrocrystallization of Rh will be discussed in the light of experiments performed on silicon, InP and GaInP. These systems are characterized by the formation of an interfacial oxide film separating electrocatalytic particles from the absorbing semiconductor. The composition and electronic structure of this latter builds a particular energetic pathway chart that defines the electrode performance.

Biography:
Marie-Christine Record has completed her PhD in 1992 from Montpellier University (France) and postdoctoral studies from Ecole Centrale Paris (France). She was an associate professor at Montpellier University from 1996 to 2004 and from 2004, she is full professor at the Aix-Marseille University (France) where she works on the elaboration of thin films materials for energy. She has published more than 80 papers in reputed journals and has been serving as an editorial board member of repute
Abstract:
Electrochemical deposition may provide an alternative process to physical deposition methods for peparing semiconducting thin films. Indeed by contrast to other methods, electrochemical deposition is a low cost, room temperature production technique, which works without vacuum atmosphere and allows one to cover substrates with complex shapes. The shortcoming of electrochemical deposition is limited control on the size, stoichiometry and crystallinity of deposits.The electrochemical atomic layer deposition (EC-ALD) method was put forward in 1991 by Gregory and Stickney. This method is based on the alternate underpotential deposition (UPD) of atomic layers of the elements to make up a compound, combining advantageously the technique of electrochemical deposition and that of atomic layer deposition (ALD). Underpotential deposition is a surface-limited electrochemical phenomenon, which makes the deposition generally limited to an atomic layer. In every cycle one monolayer of the compound is obtained, and the thickness of the deposit will only depend on the number of cycles. Each cycle consists in a series of individual steps, and each step can be optimized independently, resulting in well-controlled deposits. In the first part of this talk, I will describe the EC-ALD method and I will expose the advantages of this method compared to those of physical ones of ALD type and compared to those of other electrochemical methods. Then, I will detail the EC-ALD experiment platform designed and constructed in our group. Finally, I will present some results we got with this equipment for the electrodeposition of CoSb3 and Sb2Se3 compounds.

Biography:
Dr. Xiangyu Zhao has his expertise in electrochemical energy storage including electrochemical hydrogen storage and rechargeable batteries such as chloride ion batteries, lithium ion batteries and magnesium batteries. He received his PhD in Materials Science at the Nanjing Tech University in 2010 and then joined the same university. Meanwhile, from 1/2012 to 12/2013, he was awarded by the Guest Scientist Fellowship and worked at Karlsruhe Institute of Technology. He is (co-)author of 9 patent applications and has more than 60 papers published in international journals such as Angew Chem Int Ed, Adv Energy Mater and Energy Environ Sci.
Abstract:
Alternative battery chemistries beyond lithium ion and using abundant electrode materials have been developed. Chloride ion battery (CIB) is a new rechargeable battery based on Cl- anion transfer. This battery shows a variety of electrochemical couples with theoretical energy densities up to 2500 Wh l-1, which is superior to those of conventional lithium ion batteries. Abundant material resources such as Mg, Ca, Na and metal chlorides (e.g., FeCl3, CuCl2 and MgCl2) can be sustainable electrode candidates. The CIB includes a metal chloride/metal electrochemical couple and an ionic liquid electrolyte allowing chloride ion transfer, as reported in the proof-of-principle study of CIB operated at room temperature. The problem is that the metal chloride cathode can react with chloride ions in the electrolyte, leading to the formation of soluble complex anion. This electrode dissolution and the subsequent shuttle would limit the use of metal chloride cathode in the liquid electrolyte system. Metal oxychlorides with higher stability have been proved to be new cathode materials for CIBs. Metal oxychloride/metal systems could also show high theoretical energy densities during the chloride ion transfer. By carbon incorporation in the cathode or optimization of electrolyte composition, more than 70% of the theoretical discharge capacity of single-electron cathode such as FeOCl or VOCl could be delivered. A preliminary study on the multi-electron VOCl2 cathode was also reported in the electrode system using VOCl as cathode and Mg/MgCl2 composite as anode. Besides inorganic electrode materials in rechargeable batteries, organic electrode materials, in particular polymers, have been attracting much attention, due to their advantages of good electrochemical performance, high stability, abundant chemical elements, structural tunability and designing flexibility. Chloride ion doped polymer materilals have been studied and developed as new cathodes for chloride ion batteries. Reversible reversible redox reactions and superior cycling stability were obtained


Biography:
Oliver Clemens has obtained his PhD in 2012 years from Saarland University, Germany, followed by postdoctoral studies at the University of Birmingham, UK. He is group leader of the Joint Research Laboratory Nanomaterials, TU Darmstadt and KIT, Germany since May 2013. At the beginning of 2017 he became a junior professor (Qualifikationsprofessor) within the Materials Science Department at the TU Darmstadt, Germany within an Emmy Noether Fellowship from the German Research Foundation. He has published more than 36 papers (November 2016) in reputed journals.
Abstract:
Building batteries based on a shuttle of fluoride ions is of interest due to the high stability of fluoride as a charge carrier. Therefore, wide potential windows can be accessible in such battery systems allowing for high energy densities. So far, fluoride ion batteries (FIBs) are mainly fabricated as all solid state batteries using conversion based electrodes. However, conversion reactions are well-known to result in limited battery lifetime due to the large volume changes which arise during the cycling of the battery. In addition, high overpotentials (crystallization, charge transfer) can have a tremendous impact on battery kinetics. To improve battery kinetics, we aim to develop intercalation based fluoride ion batteries with high capacities and high voltage. In this respect, perovskite and perovskite related compounds (e. g. Ruddlesdon-Popper type structures) will be shown to serve as host lattices which can intercalate/deintercalate large amounts of fluoride ions, with theoretical capacities reaching ~ 130 mAh/g and being comparable to lithium ion battery systems. We highlight that such materials can outperform CuF2 as a high voltage cathode in agreement with by DFT based calculations. Latest developments on the identification of suitable anode materials will be described in addition to an overview of current limitations of anion based battery systems. Finallly, we will elucidate structure chemical factors which enable selectivity for the intercalating species for cation (e. g. Li and Na) in comparison to anion based batteries.
Jian Zhang
TU Dresden, Germany
Title: Efficient Hydrogen Production on MoNi4 Electrocatalysts with Fast Water Dissociation Kinetics

Biography:
Dr. Jian Zhang received his PhD degree from the institute of coal chemistry, University of Chinese Academy of Science in 2014. He worked as a post-doctor from Oct. 2014 to Jan. 2015. Since February 2015, he joined Professor Feng's group as a post-doctor in Center for Advancing Electronics Dresden (cfaed) & Department of Chemistry and Food Chemistry, Technische Universitaet Dresden. He has his expertise in developing novel electrocatalysts for water splitting and oxygen reduction by engineering the interfaces and electronic properties of active sites.
Abstract:
Large scale and sustainable production of hydrogen from water using the efficient and cost-effective electrocatalytic/photocatalytic/photoelectrocatalytic water splitting devices, e.g., water-alkali electrolyzers, is greatly promising for the future hydrogen economy. To this end, efficient, durable, and low-cost electrocatalysts are required to reduce the kinetic overpotentials of hydrogen evolution reaction (HER). Noble metal platinum (Pt) has been recognized as the most active and robust HER electrocatalyst with a near-zero onset overpotential and a high anodic current density. Unfortunately, the large-scale employment of Pt catalysts for hydrogen production is severely limited by its scarcity and high cost. For the HER in an alkaline solution, the kinetic process involves two steps: the prior water dissociation (Volmer step) and the subsequent combination of the adsorbed hydrogen (H*) into molecular hydrogen. Thus, once an electrocatalyst facilitates the initial water dissociation step on the surface, the HER performance will be improved. In this regard, we developed and design the novel electrocatalysts through engineering active sites for the water dissociation. For example, we demonstrate a novel out-diffusion strategy for synthesizing MoNi4 electrocatalysts, which can efficiently speed up the sluggish the Volmer step of the HER process in alkaline solution. The computational and experimental results reveal the fact that the kinetic energy barrier of the initial Volmer step is substantially reduced on the MoNi4 electrocatalysts. The as-constructed MoNi4 electrocatalysts supported by MoO2 cuboids exhibited an excellent electrocatalytic HER activity in 1 M KOH aqueous solution with an extremely low overpotential of ~15 mV at a current density of 10 mA cm-2 and a low Tafel slope of 30 mV decade-1, which are highly comparable to the results for the Pt and superior to those for state-of-the-art Pt-free electrocatalysts. Benefiting from its scalable preparation and excellent stability, the developed MoNi4 electrocatalyst is highly promising for practical water-alkali electrolyzer.
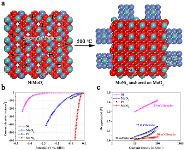
Akiyoshi Manabe
De Nora Permelec Ltd., Japan
Title: Study on performance of alkaline water electrolysis (AWE)

Biography:
Akiyoshi Manabe is General Manager of Water Electrolysis & Energy PJ team in De Nora Permelec Ltd. He finished master degree of Tokyo Institute of Technology. After that, he has developed a new development project at the forefront by Chlorine Engineers corp. Ltd., a brother company of current company. His main development of technology is as follows, activated cathode coating (low hydrogen overvoltage) for chlor-alkaline plan cell (1977-1981), Supercritical extraction of CO2 gas
Abstract:
Global warming caused by CO2 gas can no longer be ignored. Therefore, we are trying to contribute to reducing the problem with our electrolytic technology accumulated so far in our company. Our target is to develop a large AWE plant with high performance and contribute to problem solving. The performance of AWE is largely classified and influenced by four factors of anode, cathode, separator and cell structure of electrolyzer. Here, the results of electrodes and separators are mainly explained in our evaluation. Considering the use of renewable energy as standard, electricity always fluctuates in the operation of AWE. Therefore, the components of the cell must be sufficient resistant to such fluctuations. Electrode: There are two types of activated coating to reduce the overvoltage of electrode. Our investigation revealed that the anode coating of thermal decomposition is not enough tough, but the dispersion electroplating such as Raney Ni showed good durability against 100 times shutdown. During the shutdown of operation, revers current pass through in the cell. The revers current deteriorates the electrode performance and the phenomena causes difficult for anode coating life. Each saving of anode oxygen overvoltage of thermal and electroplating is around 50 mV and 100 mV compared with bare Ni. Separator: In the AWE, electrolyte is the same in both anode chamber and cathode chamber, so that diaphragm instead of ion exchange membrane can be used as separator. The point of its performance is that low cell voltage and high purity gas can be obtained. Currently, AGFA and KHI diaphragm are considered to be applicable to large-scale AWE plants. The performance of our AWE plant was around1.8 V at 5 kA/m2., 80 deg. C. Its performance is affected by the electrode to be used. The differences in cell voltage occur from 100 mV to 200 mV.

Biography:
Yu-Wu Zhong obtained his Ph.D. in July 2004 from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences (CAS) under the supervision of Prof. Guo-Qiang Lin. From September 2004 to September 2006, he has been a postdoctoral researcher at the University of Tokyo with Prof. Eiichi Nakamura. From October 2006 to September 2009, he worked with Prof Hector D. Abruna at Cornell University as a postdoctoral researcher. In October 2009, he was awarded by the “100 talent” program of CAS and joined Key Laboratory of Photochemistry of the Institute of Chemistry, CAS, to start his independent career. His research interests focus on electro-active and photofunctional organic materials and transition-metal complexes. He has published more that 110 peer-reviewed papers to date. He is currently an associate editor of RSC Advances and editorial board member of Scientific Reports and Science China Chemistry.
Abstract:
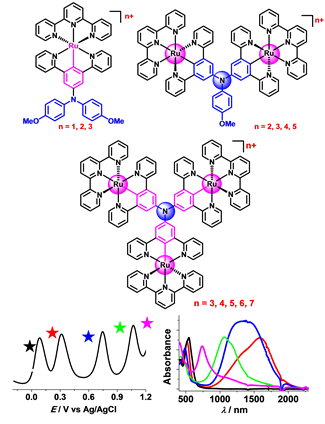
Electro-active and electrochromic materials have received a wide range of applications. The incorporation of a metal ion gives rise to new functions that are not present in common organic materials. However, the applications of electro-active organometallic materials are often hampered by their high redox potentials and difficulty in film formation. Recently, we have been interested in the design and construction of electro-active systems with cyclometalated ruthenium and triarylamine as the charge-bearing sites. These materials show strong electronic coupling and multiple reversible redox processes in low positive potential window. After modified with vinyl or triphenyl units, these can be smoothly deposited onto ITO electrode surfaces by in situ electropolymerization. The obtained films exhibit multistate NIR electrochromism with long retention time, good contrast ratio, and low switching potential. The response is about a few seconds. Depending on the number of redox sites, 3 - 5 step redox switching were realized. These films have been used for the demonstrations of flip-flop or flip-flap-flop memory with electrochemical potentials as input signals and absorbance at different wavelengths as output signals. In addition to electropolymerized films, self-assembled monolayer films of these complexes were obtained and they are useful in molecular-scale NIR electrochromism and electrochemically-gated single molecular conductance.

Biography:
Elise Deunf received her PhD in physical and analytical chemistry in 2010 under Dr Christian Amatore from the Ecole Normale Supérieure (Paris). She then joined the Lawrence Berkeley National Laboratory and University of California Berkeley as a postdoctoral fellow to develop new organic hydrogen carriers for PEM-fuel cells in collaboration with GE Global research. In 2013, she moved back to France to work with Prof. Philippe Poizot at the Institute of Materials Jean Rouxel (Nantes). Her current research focuses on molecular electrochemistry and the development of organic electrode materials for alternative and low-polluting batteries.
Abstract:
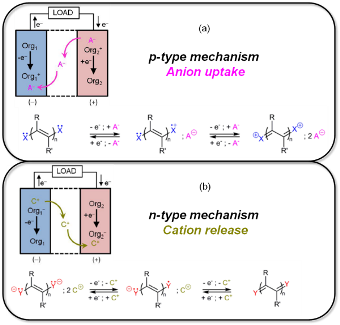
Routine access to power sources is an essential factor for developing our technology-oriented society and ensuring a better quality of life. In this context, while the implementation of renewable energy sources is in progress, electrical energy storage (EES) systems are set to play a central and potentially critical role in the next-generation energy infrastructure. Faced with a worldwide demand for powering electrified vehicles and electronic devices of all kinds, accelerated progress and innovation in the development of new and potentially “greener” electrochemical storage solutions are thus imperative. Based on the tailoring of naturally abundant chemical elements (C, H, N, O, S, in particular), organic chemistry provides great opportunities for finding innovative electrode materials able to operate in aqueous or nonaqueous electrolytes.1 Along this line, significant progress has been achieved these last ten years on redox-active organic compounds, bringing them to the attention of the energy storage community. Indeed, organic chemistry provides great opportunities for discovering innovative electrode materials able to operate both in aqueous and nonaqueous electrolytes. Additionally, it must be pointed out that two types of electrochemical insertion mechanisms can be used in practice with either reversible uptake/release of cations or anions (figure 1a, 1b respectively).2 In this communication, we will present recent advances on organic host materials and will particularly report on small organic compounds based on aromatic core structures that are functionalized by redox-active groups such as enolate and amine, and which able to reversibly host cations or anions by electrochemical reaction.3-6
Sylvio Indris
Karlsruhe Institute of Technology, Institute for Applied Materials – Energy Storage Systems, Germany
Title: Electrodes and Solid Electrolytes for Li-Ion Batteries: Local Structures, Li-Ion Mobility, and Lithiation Mechanisms

Biography:
Sylvio Indris is investigating materials that can be used as electrodes and electrolytes in Li-ion batteries. These materials are prepared by solvothermal/hydrothermal methods, sol-gel techniques, and solid-state reactions. The changes in these materials during operation in these batteries are investigated by diffraction techniques as well as X-ray absorption spectroscopy, solid-state NMR, and Mössbauer spectroscopy. Finally, the results of these investigations are used to optimize the performance of the complete Li-ion battery systems.
Abstract:
Li-ion batteries are used extensively in mobile electronic devices and in electric vehicles due to their high energy and power densities. The performance of these storage systems strongly depends on the materials used inside these batteries as electrodes and electrolytes. In order to improve these batteries, it is important to understand the fundamental diffusion mechanisms and also the fundamental electrochemical reaction mechanisms that occur during charging and discharging of the batteries, i.e. during lithiation and delithiation of the electrodes, and that are responsible for function and degradation of these systems.The changes that occur in the electrode materials during electrochemical cycling are investigated by in situ techniques including diffraction as well as spectroscopic methods [1-3]. These measurements are performed on olivine materials LiMPO4 (M = Fe, Mn, Co) and high-voltage spinel materials LiM’M’’O4 (M’, M’’ = Mn, Co, Ni, Fe) and they reveal the details of the lithiation/delithiation mechanisms and how these are related to the performance of these materials in the batteries. Solid electrolytes might offer an enhanced safety for these systems. Different oxidic, sulfidic, and polymer systems are investigated with respect to Li+ ion mobility by 7Li NMR relaxometry and field-gradient NMR spectroscopy [4-6]. From this, microscopic diffusion parameters such as the average Li ion jump rate t-1 and the activation barrier EA for single Li ion jumps can be extracted, but also the macroscopic transport of the Li ions can be observed. The results of these measurements are compared to those obtained by impedance spectroscopy.
Yan Jiao
The University of Adelaide, South Australia
Title: Evaluation of doped graphene materials for electrochemical hydrogen evolution reaction and oxygen reduction reaction

Biography:
Dr Yan Jiao's research interests involve discovering the origin of electrocatalytic activity possessed by carbon-based materials by computational chemistry. She also aims to design novel carbon-based catalysts for clean energy conversion reactions, including hydrogen evolution reaction, oxygen reduction reaction and carbon dioxide reduction reaction. She obtained her PhD in Chemical Engineering from the University of Queensland, and is currently working as a postdoctoral researcher in the University of Adelaide (UoA). She is the receiver of several awards, including Women's Research Excellence Award by UoA. She has received over 4400 citations and h-index is 19.
Abstract:
The dwindling supply of fossil fuels urge us to explore alternative power sources to drive our highly automotive society. Under this background, establish reliable clean and sustainable energy supplies are of great importance, and using electrochemical method to realize energy conversions hold a great promise. Among these reactions, hydrogen evolution reaction (HER) and oxygen reduction reaction (ORR) are the most studied, due to their fundamentality in electrocatalysis and their role in hydrogen production and fuel cells, respectively. Effective candidates for these two reactions are often based on noble metals, while carbon-based metal-free electrocatalysts generally demonstrate poorer activity. Here we report evaluation of a series of heteroatom-doped graphene materials as efficient HER and ORR electrocatalysts by density functional theory calculations, with the input of spectroscopic characterizations and electrochemical measurements. Results of theoretical computations are shown to be in good agreement with experimental observations regarding the intrinsic electrocatalytic activity and the reaction mechanisms for these two reactions. As a result, we establish volcano shaped activity trends for HER and ORR on graphene-based materials, and explore their reactivity origin to guide the design of more efficient electrocatalysts. We predict that by rationally modifying particular experimentally achievable physicochemical characteristics, a practically realizable graphene-based material will have the potential to exceed the performance of the metal-based benchmarks for these two reactions.

Biography:
Qamar Abbas received his PhD degree in technical sciences from Graz University of Technology (Austria) in 2011. The same year he joined the Institute of Chemistry and Technical Electrochemistry (ICTE) at Poznan University of Technology, Poznan, Poland where he worked as a post doc in WELCOME project funded by the Fundacja na rzecz Nauki Polskiej (FNP) from 2011 to 2015. Since Jan 2016, he is assistant researcher at the ICTE and his research interests are related with optimization of supercapacitor performance in aqueous and organic electrolytes under testing conditions and corrosion investigations and mitigation of stainless steel type alloys in aqueous media.
Abstract:
Supercapacitors (SCs) generally use activated carbon (AC) electrodes and organic electrolytes e.g. 1 mol L-1 TEABF4 in acetonitrile due to the energy vs voltage square dependence. However, due to the flammable character of acetonitrile, environment-friendly and low cost alternatives e.g. neutral aqueous Li2SO4 (pH = 6.5-7.0) exhibiting moderate voltages up to 1.5 V in SCs have been recently proposed. Such voltage exceeding water stability of 1.23 V is due to large over-potential for di-hydrogen evolution at the negative carbon electrode caused by local downshift of pH. Lately, by introducing potassium iodide (KI) in aqueous Li2SO4, AC/AC hybrid cells operating up to 1.6 V displayed high capacitance as a result of hybridization of a battery-type positive electrode and capacitor-type negative one. The battery-type performance of the positive electrode is associated with redox reactions 2I- ↔ I2 + 2e- enhancing greatly the capacity of the positive electrode than for the negative one, C+>>C-, and using equation for capacitors in series 1/C = 1/C+ + 1/C-, capacitance of cell is equal to negative electrode capacitance.
Here, we show that hybrid capacitors in aqueous KI+Li2SO4 (pH = 6.5) using symmetric carbon configuration losses capacitance upon cycling/floating at 1.5 V. When using microporous carbons as positive and negative electrodes, the former reaches to +0.692 V vs SHE, and when implementing mesoporous electrodes, the negative electrode reaches to -0.985 V vs SHE well below the di-hydrogen evolution potential (-0.46 V vs SHE). Hence, both systems display capacitance loss under cycling/floating at 1.5 V. We implement asymmetric configuration using mesoporous carbon as positive electrode to better trap iodide species, and microporous carbon as negative one to improve hydrogen storage, to balance the system. TPD, Raman, Gas adsorption and electrochemical data on electrodes and cells (Figure 1) proves that the oxidation of positive electrode and hydrogen production on negative electrode are reduced, improving the cyclability, capacitance and energy efficiency of the cell up to 10,000 cycles.
- ms
Location: Berlin, Germany
- Advances in Electrochemistry | Theoretical and Computational Electrochemistry | Electroanalytical Chemistry | Electrochemical Energy Conversion and Storage | Bioelectrochemistry
Location: Berlin, Germany
Session Introduction
Rolando Guidelli
Professor
Title: What Ion Flow Along Ion Channels Can Tell About Their Functional Activity

Biography:
Rolando Guidelli obtained his degree in Chemistry at Florence University, Italy, and was appointed by Florence University as a lecturer in Electrochemistry. He was then promoted to full professor of Electrochemistry at the age of 32 years in the Faculty of Science of Florence University. His scientific interests have been focused on electrode kinetics, structure of metal/water interfaces, and bioelectrochemistry. He has won several distinguished prizes in the field of electrochemistry and has published more than 230 papers in reputed journals and several book chapters. Furthermore, he has served as the organizer of several conferences in the field.
Abstract:
The most direct method for verifying the functional activity of channel-forming peptides and proteins consists in monitoring the flow of physiologically relevant inorganic ions, such as Na+, K+ and Cl–, along the ion channels. Measurements of average and single channel currents across bilayer lipid membranes (BLMs) interposed between two aqueous solutions are extensively employed to this end. A major drawback of BLMs is their fragility, high sensitivity toward vibrations and mechanical shocks, and low resistance to electric fields. To overcome this problem, metal-supported tethered BLMs (tBLMs) have been devised, where the BLM is anchored to the metal via a hydrophilic spacer that replaces the water phase on the metal side. However, only mercury-supported tBLMs can measure and regulate the flow of the above inorganic ions, thanks to mercury liquid state and high hydrogen overpotential. Thus, they react to the presence of proteins, charges and physical forces in a dynamic and responsive manner, by reorganizing upon interaction with external perturbations and mimicking the functionality of living cell membranes. The potential of Hg-supported tBLMs is illustrated by the use of different electrochemical techniques. Moreover, a tBLM formed on a mercury cap electrodeposited on a platinum microdisk yields a micro tBLM that maintains the same fluidity and lipid lateral mobility as on a hanging mercury drop and allows the recording of single channel currents and of two-photon fluorescence lifetime images of lipid rafts and gel-phase microdomains, opening the way to its use in high throughput screening applications.

Biography:
Grzegorz Milczarek received his PhD and D.Sc. degree from Faculty of Chemical Technology at Poznan University of Technology (Poznan, Poland). Currently hi is a Professor at the same university. He is also a head of the Division of General and Analytical Chemistry. His research areas include: electrocatalysis, preparation of new materials for energy conversion and storage, preparation of chemically modified electrodes to be used as electrochemical sensors. He has published over 50 peer reviewed publications.
Abstract:
Lignin is an abundant biopolymer on earth, present in all woods and grasses. In its native form, it is insoluble in any solvent and therefore useless for solution chemistry, in spite of its documented antioxidant properties. On the other hand, chemical derivatives of lignin (technical lignins) are produced in the quantities of millions tons per annum as a by-product in the production of cellulose from wood or other plant materials. Depending on the technology used, technical lignins differ in molecular weight, chemical composition and the structure of chemical moieties that make them soluble in organic solvent or even in water. Of particular interest are lignosulfonates that exhibit both poly-electrolyte and poly-phenol character due to the presence of ionisable and hydrophilic sulfonic moieties and substituted phenol groups responsible for their reducing properties. In this report, we demonstrate that technical lignosulfonates can be used as both reducing and stabilizing agents for cost-effective synthesis of various nanoparticles including noble metal nanoparticles and transition metal hexacyanoferrates that show valuable multifunctional electrochemical properties. The obtained hybrid materials show multiple redox activity due to the presence of lignin-derived quinones. Therefore, such materials can be used as energy storing materials or sensing platforms for electrochemical sensors (see for example Fig. 1). Lignosulfonates and lignosulfonate-stabilized nanoparticles can be also introduced into chemically or electrochemically polymerized conducting polymers e.g. polyaniline, poly-pyrrole and poly-ethylenedioxythiophene.
Akiyoshi Manabe
De Nora Permelec Ltd., Japan
Title: Study on performance of alkaline water electrolysis (AWE)

Biography:
Akiyoshi Manabe is General Manager of Water Electrolysis & Energy PJ team in De Nora Permelec Ltd. He finished master degree of Tokyo Institute of Technology. After that, he has developed a new development project at the forefront by Chlorine Engineers corp. Ltd., a brother company of current company. His main development of technology is as follows, activated cathode coating (low hydrogen overvoltage) for chlor-alkaline plan cell (1977-1981), Supercritical extraction of CO2 gas
Abstract:
Global warming caused by CO2 gas can no longer be ignored. Therefore, we are trying to contribute to reducing the problem with our electrolytic technology accumulated so far in our company. Our target is to develop a large AWE plant with high performance and contribute to problem solving. The performance of AWE is largely classified and influenced by four factors of anode, cathode, separator and cell structure of electrolyzer. Here, the results of electrodes and separators are mainly explained in our evaluation. Considering the use of renewable energy as standard, electricity always fluctuates in the operation of AWE. Therefore, the components of the cell must be sufficient resistant to such fluctuations. Electrode: There are two types of activated coating to reduce the overvoltage of electrode. Our investigation revealed that the anode coating of thermal decomposition is not enough tough, but the dispersion electroplating such as Raney Ni showed good durability against 100 times shutdown. During the shutdown of operation, revers current pass through in the cell. The revers current deteriorates the electrode performance and the phenomena causes difficult for anode coating life. Each saving of anode oxygen overvoltage of thermal and electroplating is around 50 mV and 100 mV compared with bare Ni. Separator: In the AWE, electrolyte is the same in both anode chamber and cathode chamber, so that diaphragm instead of ion exchange membrane can be used as separator. The point of its performance is that low cell voltage and high purity gas can be obtained. Currently, AGFA and KHI diaphragm are considered to be applicable to large-scale AWE plants. The performance of our AWE plant was around1.8 V at 5 kA/m2., 80 deg. C. Its performance is affected by the electrode to be used. The differences in cell voltage occur from 100 mV to 200 mV.
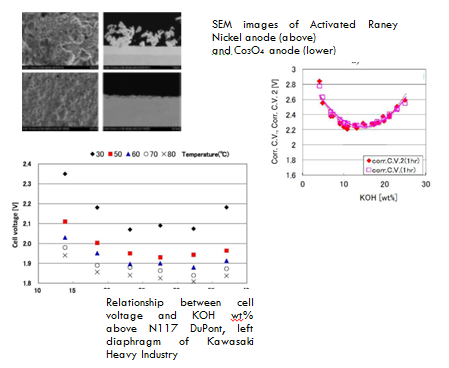

Biography:
Prof. K. Boniface Kokoh is developing electrocatalytic materials for sustainable energy conversion and storage. He manages an electrocatalysis group that prepares electrode nanomaterials that are suitable to each application. His research topics concern hydrogen production in a solid polymer electrolyte water electrolyzer, abiotic electrodes design for hybrid biofuel cells and the CO2 electroreduction to platform molecules.
Abstract:
Statement Glucose is an environmentally friendly and sustainable carbohydrate that is actually a cyclized aldehyde in aqueous solution i.e. a hemiacetal. Its selective oxidation by the anomeric carbon in C1-position without any function protection is of great interest for cogeneration because the electrochemical process enables converting this highly functionalized organic molecule into electricity, heat and added-value chemicals. For this objective, we decide to synthesize effective nanomaterials towards glucose conversion at the anode and the oxygen reduction reaction (ORR) in the cathodic side of a fuel cell. The physical characterizations of the prepared materials and their electrochemical analysis at each half-cell, permitted to optimize the activity of the electrodes according to their elemental composition and structure. Furthermore, a constructed direct glucose fuel cell (DGFC) delivered an open-circuit voltage of 1.1 V in 0.5 mol L-1 NaOH and an outstanding output power of 2 mW cm-2. Complementary analytical techniques were employed to quantify the reaction processes involved in each compartment, determine the reaction products resulted from the glucose transformation and thereby, to understand reaction mechanisms of the glucose oxidation and the ORR over the synthesized electrode materials used in alkaline medium. As results, the identification of gluconate as the sole reaction product in the anodic side showed a selective 2-electron conversion of glucose, while the ORR proceeded through a 4-electron pathway over the designed cathode catalyst

Biography:
Shozo Yanagida is an emeritus professor of Osaka University and a research director of “Research Association for Technological Innovation of Organic Photovoltaics” (RATO) of University of Tokyo. Since he was promoted to a professor of newly established “Koza” (research course) of Graduate School of Engineering in Osaka University (1980), he had contributed to photochemical conversion of solar energy, e.g., excellent photocatalyses of both nano-sized (quantized) ZnS and poly- & oligo-paraphenylene. When he was staying at SERI (now ENREL) as a visiting professor of Dr. A. Nozik’s group in 1984, he understood that organic molecules and their aggregates are kinds of quantum dots themselves. He has his expertise in evaluation of dye-sensitized solar cells, i.e., molecular structured photovoltaics, and passion in improving photo-conversion efficiency and long-term durability of solar cells in view of unidirectional electron flow and electron lifetime in solar cell devices.
Abstract:
So-called perovskite solar cells (PSC) are composed of PbI64- (MeNH3+)4 salt, where PbI64- plays an essential role as an effective solar light sensitizer with keeping semiconducting property even when aligned each other. Density-functional-theory-based molecular modeling (DFT/MM) using reported X-ray crystallographic structure of PbI64-/MeNH3+/H2O salt (named FOLLIB in Cambridge Structural Data) validates that the packing unit consisting of {(PbI64-)9[(MeNH3+)2-H2O]2(MeNH3+-H2O)2(MeNH3+)2}28- should show UV/Vis absorption spectrum at lmax=424 nm (pale yellow color) as observed for the PbI64- crystal. DFT/MM of the FOLLIB horizontal aligned component, [(PbI6 4-[(MeNH3+)2-H2O]2(MeNH3+-H2O)2(MeNH3+)2/(PbI6 4-)2)4- verifies that the component has narrow energy gap of 0.3eV, predicting excellent semiconducting property of the PbI64- alignment with MeNH3+. Three H2O-free PbI64-/MeNH3+ aligned components, PbI64-(MeNH3+)4, [PbI64-(CH3NH3+)3]- and [PbI64-(CH3NH3+)2]2- are molecular modeled and verified to have UV/Vis spectra at lmax=570nm, lmaxï€ ï€½762nm, and lmax=945nm, respectively. Mixtures of them will be colored black, which is consistent with observable black coloration of PbI64- alignments with MeNH3+ in amorphous solute state. It is further verified that PbI64- undergoes van der Waals and Coulomb interactions both with electron accepting layers, i.e., nc-TiO2 in PSC of nc-TiO2/MeNH3PbI3/spiro-OMeTAD and with electron donating layer, i.e., spiro-OMeTAD in the PSC. The molecular orbital structure and electrostatic potential map verifies formation of tight interaction between them. The electron density-based alignment PbI64- validates unidirectional electron transport at both interfaces, resulting in high open-circuited voltage (Voc) of ~1.0 eV in PSC. In addition, the semi-conducting sensitizing layer of PbI64-/MeNH3+ components validates excellent short-circuited photocurrent (Jsc), and respectable fill factor of PSC. The PbI64--aligned solar cell will be regarded as a kind of quantum dot solar cell
Andrés G. Muñoz
GRS gGmbH, Germany
Title: Progress and Challenges in Galvanic Activation of Electrodes for Solar Water Splitting

Biography:
Andrés G. Muñoz is a senior scientist passionate about fundamental questions of electroplating of semiconductors and metals. He has gained his expertise by working with renowned scientists from the German electrochemistry school at the Heinrich-Heine University of Düsseldorf and the Research Centre Jülich. After being distinguished with an Alexander von Humboldt fellowship, he succeeded in combining his wide background in electrochemistry with surface science on joining the Institute for Solar Fuels and Energy Storage Materials at the Helmholtz-Center in Berlin. He resolved fundamental questions of interfacial photoelectrochemical processes in solar energy conversion systems. Presently, his is also involved in projects concerning the application of electrodeposition to improve the corrosion
Abstract:
Solar water splitting tandem devices are a promising route for building a carbon-neutral energy infrastructure based on hydrogen as a fuel. The development horizon suggests the use of technologically advanced semiconductor materials, mainly III-V ones, due to its fast technical realization. The coupling of high-efficiency photovoltaics with electrochemical water splitting processes (two in one systems) requires from smart conditioning methods to turn semiconductor surfaces electrochemical active (figure 1). Particularly, the decoration of with noble metals, such as Pt and Rh is one of the most pursued approaches in the design of photocathodes to be integrated in tandem-type devices. Electroplating is preferred method due to its simplicity, its low cost and its power for accessing hidden places in complicated geometrical shapes (structured surfaces with enhanced photonic properties). The optical and electrocatalytic performance of deposited metal films can be enhanced by achieving a particular size and distribution of their nano-dimensioned grains. The chemical composition of the electrolytic bath, the applied potential program and illumination intensity constitute the main tools to do this.Electroplating of noble metals, however, triggers interfacial reactions generating chemical and electronic surface transformations. This is due to the complex multi-electron transfer involved in the metal phase formation and the particular high thermodynamic redox potential. Therefore, the selection of a galvanic method must balance coupled interfacial processes with desired properties of the electrocatalytic film. The complex picture of electrocrystallization of Rh will be discussed in the light of experiments performed on silicon, InP and GaInP. These systems are characterized by the formation of an interfacial oxide film separating electrocatalytic particles from the absorbing semiconductor. The composition and electronic structure of this latter builds a particular energetic pathway chart that defines the electrode performance.


Biography:
Pascal Boulet has completed his PhD in 2001 from both the University of Lyon (France) the University of Geneva (Switzerland). He then moved to University College London (England) as a postdoctoral fellow for two years. Since 2003 he is associate professor at the Aix-Marseille University (France) where he works on materials for energy using computational chemistry tools. He has published over than 50 papers in reputed journals.
Abstract:
The phenomenon of deposition of a monolayer amount of metal atoms onto a dissimilar, more noble metal electrode at a potential more anodic than expected from the Nernstian equilibrium potential was recognized about 70 years ago but it was not before 1974 that Kolb, Gerischer and Przasnyski explained theoretically this concept known as the underpotential deposition (UPD) DVupd. Using over 20 couples of metals these authors found that DVupd followed the simple, linear equation DVupd =½Df, where Df is the difference in work functions between the adsorbate and the substrate. For most of the metal couples investigated, the relation was verified. However, this seemingly oversimplified relation was soon after contradicted by Bewick and Thomas’ experiments on the deposition of Tl and Pb on Ag single crystal electrodes. Thenceforth, there has been tremendous works achieved by several authors to refine the microscopic understanding of the UPD process. These theoretical works were often accompanied by numerical simulation approaches at the atomistic level (Monte Carlo and kinetic Monte Carlo techniques) and quantum mechanical calculations. However, despite the increase of computer and softwares capabilities the models used in these studies cannot fully capture the whole complexity involved in the UPD process and simplified model systems had to be used. In this paper, we will review the microscopic theory of UPD and the results obtained from computational, atomistic approaches. We will show the assets but also the weaknesses, and the foreseeable improvements in particular in the realm of the density-functional theory.
Jian Zhang
TU Dresden, Germany
Title: Efficient Hydrogen Production on MoNi4 Electrocatalysts with Fast Water Dissociation Kinetics

Biography:
Dr. Jian Zhang received his PhD degree from the institute of coal chemistry, University of Chinese Academy of Science in 2014. He worked as a post-doctor from Oct. 2014 to Jan. 2015. Since February 2015, he joined Professor Feng's group as a post-doctor in Center for Advancing Electronics Dresden (cfaed) & Department of Chemistry and Food Chemistry, Technische Universitaet Dresden. He has his expertise in developing novel electrocatalysts for water splitting and oxygen reduction by engineering the interfaces and electronic properties of active sites.
Abstract:
Large scale and sustainable production of hydrogen from water using the efficient and cost-effective electrocatalytic/photocatalytic/photoelectrocatalytic water splitting devices, e.g., water-alkali electrolyzers, is greatly promising for the future hydrogen economy. To this end, efficient, durable, and low-cost electrocatalysts are required to reduce the kinetic overpotentials of hydrogen evolution reaction (HER). Noble metal platinum (Pt) has been recognized as the most active and robust HER electrocatalyst with a near-zero onset overpotential and a high anodic current density. Unfortunately, the large-scale employment of Pt catalysts for hydrogen production is severely limited by its scarcity and high cost.
For the HER in an alkaline solution, the kinetic process involves two steps: the prior water dissociation (Volmer step) and the subsequent combination of the adsorbed hydrogen (H*) into molecular hydrogen. Thus, once an electrocatalyst facilitates the initial water dissociation step on the surface, the HER performance will be improved. In this regard, we developed and design the novel electrocatalysts through engineering active sites for the water dissociation. For example, we demonstrate a novel out-diffusion strategy for synthesizing MoNi4 electrocatalysts, which can efficiently speed up the sluggish the Volmer step of the HER process in alkaline solution. The computational and experimental results reveal the fact that the kinetic energy barrier of the initial Volmer step is substantially reduced on the MoNi4 electrocatalysts. The as-constructed MoNi4 electrocatalysts supported by MoO2 cuboids exhibited an excellent electrocatalytic HER activity in 1 M KOH aqueous solution with an extremely low overpotential of ~15 mV at a current density of 10 mA cm-2 and a low Tafel slope of 30 mV decade-1, which are highly comparable to the results for the Pt and superior to those for state-of-the-art Pt-free electrocatalysts. Benefiting from its scalable preparation and excellent stability, the developed MoNi4 electrocatalyst is highly promising for practical water-alkali electrolyzer.

Qamar Abbas
Poznan University of Technology, Poland
Title: Iodides confinement within activated carbon porosity resulting battery-type electrode for aqueous hybrid supercapacitor

Biography:
Qamar Abbas received his PhD degree in technical sciences from Graz University of Technology (Austria) in 2011. The same year he joined the Institute of Chemistry and Technical Electrochemistry (ICTE) at Poznan University of Technology, Poznan, Poland where he worked as a post doc in WELCOME project funded by the Fundacja na rzecz Nauki Polskiej (FNP) from 2011 to 2015. Since Jan 2016, he is assistant researcher at the ICTE and his research interests are related with optimization of supercapacitor performance in aqueous and organic electrolytes under testing conditions and corrosion investigations and mitigation of stainless steel type alloys in aqueous media.
Abstract:
Supercapacitors (SCs) generally use activated carbon (AC) electrodes and organic electrolytes e.g. 1 mol L-1 TEABF4 in acetonitrile due to the energy vs voltage square dependence. However, due to the flammable character of acetonitrile, environment-friendly and low cost alternatives e.g. neutral aqueous Li2SO4 (pH = 6.5-7.0) exhibiting moderate voltages up to 1.5 V in SCs have been recently proposed. Such voltage exceeding water stability of 1.23 V is due to large over-potential for di-hydrogen evolution at the negative carbon electrode caused by local downshift of pH. Lately, by introducing potassium iodide (KI) in aqueous Li2SO4, AC/AC hybrid cells operating up to 1.6 V displayed high capacitance as a result of hybridization of a battery-type positive electrode and capacitor-type negative one. The battery-type performance of the positive electrode is associated with redox reactions 2I- ↔ I2 + 2e- enhancing greatly the capacity of the positive electrode than for the negative one, C+>>C-, and using equation for capacitors in series 1/C = 1/C+ + 1/C-, capacitance of cell is equal to negative electrode capacitance.
Here, we show that hybrid capacitors in aqueous KI+Li2SO4 (pH = 6.5) using symmetric carbon configuration losses capacitance upon cycling/floating at 1.5 V. When using microporous carbons as positive and negative electrodes, the former reaches to +0.692 V vs SHE, and when implementing mesoporous electrodes, the negative electrode reaches to -0.985 V vs SHE well below the di-hydrogen evolution potential (-0.46 V vs SHE). Hence, both systems display capacitance loss under cycling/floating at 1.5 V. We implement asymmetric configuration using mesoporous carbon as positive electrode to better trap iodide species, and microporous carbon as negative one to improve hydrogen storage, to balance the system. TPD, Raman, Gas adsorption and electrochemical data on electrodes and cells (Figure 1) proves that the oxidation of positive electrode and hydrogen production on negative electrode are reduced, improving the cyclability, capacitance and energy efficiency of the cell up to 10,000 cycles.

Ik-Soo Shin
Soongsil University, Republic of Korea
Title: Electrochemiluminescent Chemodosimetric Sensor for Rapid Determination of cyanide

Biography:
Dr. Ik-Soo Shin was born in 1975 in Korea, and obtained Ph.D. degree from Seoul National University (SNU) in 2007. He was a postdoctoral fellow at the University of Texas at Austin, and SNU. Currently he is an assistant professor at Soongsil University, in Korea. His research interests include the design and development of biosensors based on electrochemical method.
Abstract:
Recently, the use of electrochemiluminescence (ECL) has drawn great attention due to its advantages such as high sensitivity, wide linear response range, good reproducibility and easy operation. However, ECL sensing of small molecules is still a great challenge because most of ECL detection methods have been developed via specific biomacromolecular recognition such as antibody-antigen and aptamer-protein interactions. In the talk, I will introduce about one of recent researches, the ECL chemodosimeter for the rapid determination of cyanider. A organometallic iridium(III) complex (1) with phenyl isoquinoline main ligands is rationally designed and synthesized for the ECL “turn-on” molecular probe, and the dicyanovinyl branch attached to the end of the main ligand plays as a role of recognition site for cyanide. Upon the reaction with cyanide in aqueous solution, 1 displays drastically enhanced ECL emission with widely linear correlation between 0-0.40 mM of cyanide when the concentration of 1 is fixed to 10 uM. The method can be successfully applied in direct determination of cyanide in tab water, and the result shows that it is versatile enough for the rapid point-of-care determination of cyanide in the presence of interferences even with high reliability and reproducibility. Theoretical studies were carried out for the rational design of the probe by predicting its HOMO/LUMO energy levels as well as electronic distributions. This ECL-based chemodosimetric sensor suggests a new and versatile analysis platform for rapid determination of molecular toxins in real samples

Biography:
Shijian CHEN is a professor at Chongqing University. His research areas are wide-band-gap semiconductor based materials and devices; new energy materials and material design from the first-principles computational. His current research specifically focuses on new materials for production of hydrogen fuel from water. Before he joined Chongqing University, he was an Australian Postdoctoral Fellow (APD) at Monash University from 2008 to 2012, and as an Alexander von Humboldt research fellow at University of Hannover from 2007-2008. He is the author of over 50 peer-reviewed publications with more than 1000 citations. He received PH.D at Chinese Academy of Sciences at 2006.
Abstract:
The design and development of high-efficiency and non-noble metal hydrogen evolution reaction (HER) electrocatalysts with optimized nanostructures for human clean and sustainable energy systems has attracted significant research interest over the past years. Herein, self-supported transition-metals poly-phosphides (TMP) (such as CoP3, WP2) nanoneedle arrays on carbon cloth were topotactically fabricated by in situ phosphidation of a transition-metals oxides nanoneedle arrays precursor. Such a binder-free flexible HER electrocatalysts with integrated three-dimensional nanostructures can not only provide a large surface area to expose abundant active sites, but also facilitate electrolyte penetration for electrons and electrolyte ions, which exhibit superior bifunctional electrocatalytic activity and durability for both the HER and OER. In addition, density functional theory (DFT) calculations indicate a low kinetic energy barrier for H atom adsorption on the TMP surface which guarantees the excellent catalytic activity of the catalyst. The excellent electrocatalytic activity makes the present 3D structured TMP electrocatalysts promising catalysts for large scale highly pure hydrogen evolution by electrochemical water splitting.
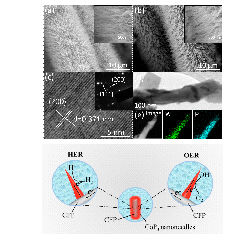

Biography:
Dr. Amedeo Capobianco earned his PhD in 2008 at University of Salerno, where he now works as an assistant professor. His current research interests include the theoretical treatment of charge transport phenomena in biochemical systems and in organic materials.
Abstract:
Singly ionized DNA exhibits long range hole transport (HT) covering distances up to 200 Å [1]. Hole transport has great biochemical and technological relevance, as it controls the site distribution of the oxidative damage in DNA and makes DNA a potential conduit for nanoelectronics, used both as template and as active component. Different mechanisms for the hole transport have been invoked so far: ion gated transport, thermal activated hopping, and superexchange among others. Independent of the kinetic mechanism, the of hole transfer in oxidized DNA is modulated by the complex mosaic of low-lying electronic states, whose accessibility depends on the effective in-situ hole energies of nucleobases and the electronic couplings between interacting nucleobases.The hole energies for adenine (A) and guanine sites were previously quantified by using voltammetry, the influence of hydrogen bonds on the oxidation potential was also determined. Information for cytosine oxidation was inferred by spectroelectrochemistry experiments.Here we have focused our attention on electronic coupling parameters. The latter have been estimated by the lowering of the oxidation potential observed for DNA oligonucleotides containing an increasing number of stacked homo-purine nucleobases. Voltammetric data were interpreted in terms of the two state model. Experimental results were further confirmed by theoretical computations carried out at the full quantum level for single and double stranded oligonucleotides including sugar-phosphate backbone and solvation.The observed lowering of the oxidation potential for single stranded oligonucleotides containing an increasing number of adjacent adenines amounts to ca 0.3 V for the AA step, in good agreement with theoretical predictions. That result suggests that adenine tracts give rise to delocalized hole domains (Figure 1), greatly facilitating hole transport in DNA . Although stacking interactions are found to be less effective in stabilizing the positive charge on adjacent guanines inside a strand, our results confirm that guanine tracts constitute a very efficient trap for the hole .

Biography:
Ingo Krossing studied chemistry in Munich (LMU) and finished his Ph.D. thesis 1997 (with Prof. H. Nöth). From 1997 to 1999, he worked as Feodor Lynen postdoc with Prof. J. Passmore at UNB, Canada. In 1999, he started his independent career as a Liebig- and DFG-Heisenberg-Fellow at the Universität Karlsruhe (TH) (mentor: Prof. H. Schnöckel). 2004 he changed as assistant professor to the Ecole Polytechnic Federale de Lausanne (EPFL), before being appointed Chair of Inorganic Chemistry at the Albert-Ludwigs-Universität Freiburg in 2006. His research interests cover ionic systems from reactive cations to ionic liquids, as well as electrochemical energy storage. With an ongoing ERC Advanced Grant he develops absolute acidity and reducity scales.
Abstract:
The comparison of acid-base and redox chemistry in their acidity (=> protonation) and reducity (=> electronation) scales is currently limited to measurements within one homogenous medium / solvent. Yet, it is crucial being able to measure chemical potentials of the proton and electron over medium boundaries to overcome this limit. Thermodynamically, the chemical potential differences of proton and electron are straightforward to describe, given the unified reference states ideal proton gas (1 bar) and electron gas (1 bar) we suggested in 2010 (proton gas)[1] and 2014 (electron gas).[2] This unifying concept was developed as part of our in March 2017 ceasing ERC UniChem project. The Protoelectric Potential Map PPM: In analogy to the proto- and electrochemical window of one medium like water, the PPM is a 2D plot of the medium independent pHabs vs. peabs values based on these unifying reference states. Any reaction with transfer of protons and / or electrons in any medium may be placed on the PPM. Given that the respective transfer energies are known , the thermodynamic relations between them may directly be compared over phase and medium boundaries.
This plot reveals at a glance the differences of reducity (i.e. electronation power, synonymous to redox potential) of a redox system (marked by colors) in dependence of the solvent S (marked by digits). For example, the Pearson soft deelectronator Ag+ (green) is up to about 3.5 V or 350 kJ mol–1 more effective in hard solvents like HF (2) or DCE (7) rather than in soft media like NH3 (3) or the Ionic Liquid (IL) HMIM Br (8).


Biography:
Milko van der Boom completed his B.Sc. in Chemical Engineering at the Amsterdam University of Applied Sciences and his M.Sc. degree in Inorganic Chemistry at the University of Amsterdam. In 1994, he enrolled as a doctoral student at the Weizmann Institute of Science, where he studied Organometallic Chemistry; he was awarded his Ph.D. degree with distinction in 1999. After three years of postdoctoral work at Northwestern University, where he studied the formation of functional organic films, he returned as a faculty member to the Weizmann Institute’s Department of Organic Chemistry. His interdisciplinary materials chemistry research focuses on metalloorganic-oriented synthetic and mechanistic studies. Prof. van der Boom's prizes and honors include an Alon Fellowship from the Israel Council for Higher Education, the Henri Gutwirth Prize from the Technion, and the Israel Chemistry Society's Prize for Excellent Young Chemists. He is the first incumbent of the Bruce A. Pearlman Professional Chair.
Abstract:
Directional electron-transfer events are the basis of many technologically important systems and biological processes. In this study, we demonstrate how the distance over which electron transfer occurs through organic materials can be controlled and extended. Coating of conductive surfaces with nanoscale layers of redox-active metal complexes allows the electrochemical addressing of additional but distant layers that are otherwise electrochemically silent. We also show that our composite materials can pass electrons selectively in directions that are determined by the positioning of redox-active metal complexes and the distances between them. These electron-transfer processes can be made dominantly uni- or bidirectional. Our design strategy involves 1) a set of isostructurally well-defined metal complexes with different electron affinities, 2) a scalable metal-organic spacer, and 3) a versatile assembly approach that allows systematic variation of material composition, structure, and electron transfer properties. We control the electrochemical communication between interfaces by the deposition sequence of the components and the length of the spacer, and therefore we are able to program the bulk properties of the assemblies.

Biography:
Mitsuharu Tabuchi is an inorganic synthetic chemist using wet-chemical process for oxide-based positive electrode material of lithium-ion battery. His synthetic experiences above 15 years were constructed mainly to get homogeneous LiFeO2-Li2MnO3 solid solution. The constructed synthetic route is also effective for preparing LiNi1/2Mn1/2O2-Li2MnO3 one. He has also characterization experience using 57Fe Mössbauer spectroscopy and X-ray Rietveld analysis to know charge compensation mechanism for LiFeO2-Li2MnO3 system. He found also Fe and Ni substituted Li2MnO3 and it has high-potential for practical use, because it can be prepared largely by chemical company (Tanaka Chemical Corporation) and then is electrochemically tested by battery manufacturer (NEC Corporation) as power-source of electronic vehicle (EV).
Abstract:
Li-excess positive electrode material (LiMO2-Li2MnO3 solid solution, M=Ni1/2Mn1/2 or Ni1/3Mn1/3Co1/3) is a good candidate as its high-capacity (250 mAh/g) and acceptable discharge voltage above 3.5 V for high energy-density lithium-ion battery to EV and PHEV application. Especially LiNi1/2Mn1/2O2-Li2MnO3 solid solution (Li1+x(NiyMn1-y)1-xO2, 0

Biography:
Dr. Ruiyong Chen received his Ph.D. degree (summa cum laude) in 2011 in physical chemistry from the Saarland University, Saarbrücken, Germany. He is currently a habilitation candidate at the Saarland University and a project leader at the KIST Europe. He has expertise in synthesis and characterization of electrode materials for energy storage systems and energy-efficient electrolysis. He has developed new two lithium intercalation cathode materials with disordered rock-salt structure for improving reversible capacity (up to 400 mAh g-1) for lithium ion batteries. He is currently developing new high voltage aqueous electrolytes for stationary energy storage using redox flow batteries.
Abstract:
Redox flow batteries are promising for large-scale energy storage applications due to their economy and scalability, in comparison with other energy storage systems. In addition, redox flow batteries have unique character of decoupled energy storage and power generation capability. Aqueous redox flow battery systems may offer safe operation, low cost and fast ionic mobility (power generation) in comparison with non-aqueous systems. However, due to the limits in the electrochemical stability window of water, aqueous systems can generally only be operated with low cell voltage (< 1.5 V). Conventional aqueous flow batteries have thus low energy density (< 30 Wh L-1), which is far below that for Li-ion batteries. Non-aqueous electrolytes using organic solvents, although can offer wide electrochemical window, suffer from low ionic conductivity, low solubility for active species and safety concerns. To access high cell voltage in aqueous systems, Chen et al., have recently developed new aqueous ionic liquid electrolytes with widened electrochemical window of about 3 V, using a concentrated aqueous solution of 1-butyl-3-methylimidazolium chloride (BMImCl). Herein, we explore various ionic liquid-based electrolytes and compatible redox couples (soluble species and semi-solid suspension utilizing solid-state cation/anion intercalation reactions) in the attempt to obtain redox flow batteries with high cell voltage and high energy density.

Biography:
Oliver Clemens has obtained his PhD in 2012 years from Saarland University, Germany, followed by postdoctoral studies at the University of Birmingham, UK. He is group leader of the Joint Research Laboratory Nanomaterials, TU Darmstadt and KIT, Germany since May 2013. At the beginning of 2017 he became a junior professor (Qualifikationsprofessor) within the Materials Science Department at the TU Darmstadt, Germany within an Emmy Noether Fellowship from the German Research Foundation. He has published more than 36 papers (November 2016) in reputed journals.
Abstract:
Building batteries based on a shuttle of fluoride ions is of interest due to the high stability of fluoride as a charge carrier. Therefore, wide potential windows can be accessible in such battery systems allowing for high energy densities. So far, fluoride ion batteries (FIBs) are mainly fabricated as all solid state batteries using conversion based electrodes. However, conversion reactions are well-known to result in limited battery lifetime due to the large volume changes which arise during the cycling of the battery. In addition, high overpotentials (crystallization, charge transfer) can have a tremendous impact on battery kinetics.To improve battery kinetics, we aim to develop intercalation based fluoride ion batteries with high capacities and high voltage. In this respect, perovskite and perovskite related compounds (e. g. Ruddlesdon-Popper type structures) will be shown to serve as host lattices which can intercalate/deintercalate large amounts of fluoride ions, with theoretical capacities reaching ~ 130 mAh/g and being comparable to lithium ion battery systems. We highlight that such materials can outperform CuF2 as a high voltage cathode in agreement with by DFT based calculations. Latest developments on the identification of suitable anode materials will be described in addition to an overview of current limitations of anion based battery systems. Finallly, we will elucidate structure chemical factors which enable selectivity for the intercalating species for cation (e. g. Li and Na) in comparison to anion based batteries.

Biography:
Dr. Teko W. Napporn is a Researcher of French National Center for Scientific Research (CNRS) and also Adjunct professor at the Institute of Advanced Sciences of Yokohama National University. He is developing nanostructured electrocatalysts for energy conversion (Fuel cells) and storage (batteries and water electrolysis). His research topics deal with the surface structural effect in electrochemistry at electrified Solid-Liquid Interfaces, hybrid biofuel cells. He is also involved in the development of single chamber solid oxide fuel cell
Abstract:
Gold at nanoscale fascinated scientists who developed important research topics over physics, chemistry, medicine and biotechnology. The last three decades, various innovative investigations emerged for determining the role of size, the morphology on the unexpected properties observed for gold at nanoscale. In electrocatalysis, gold nanoparticles (AuNPs) were often used to understand the intrinsic relationship between their size, morphology and activity towards two main reactions: (i) the electrooxidation of organic molecules that have a great interest as fuel in fuel cell applications, and (ii) the oxygen reduction reaction (ORR). Electrocatalysis investigations on support-less gold nanorods have shown that it is a challenge to study the intrinsic properties of their surface through their interaction with a reactive molecule. Our recent results on gold nanospheres revealed that the size of these nanomaterials plays a key role in their electrochemical response. Therefore spherical gold nanoparticles (AuNSs) with a mean diameter from 4 to 15 nm were successfully synthesized. UV-visible spectroscopy, Transmission electron microscopy, and Under-Potential Deposition of lead (UPD) were used for determining their morphology, size and approaching their surface crystallographic structure. UPD of lead reveals that their crystallographic facets are affected by their size and the growth process. In alkaline medium, the oxidation of glucose was used to evaluate their electroactivity. As results, small AuNSs exhibited drastic increase of catalytic activity (fig. 1). This feature might result in the high specific area and reactivity of the surface electron induced by their small size. The study of the reaction mechanism was investigated by in situ Fourier transform infrared reflectance spectroscopy. Gluconolactone and gluconate were identified respectively as the intermediate and the final reaction product of the glucose electrooxidation.
Atsushi Kitada
Kyoto University, Japan
Title: Electrochemical properties of AlCl3/glyme baths for room temperature aluminum electrodeposition

Biography:
Dr. Atsushi Kitada holds an assistant professorship for Department of Materials Science and Engineering at Kyoto University, Japan, since 2012. His main research topics are focused on synthesis and preparation of liquid and solid materials with electrochemical, electronic, and magnetic properties. He focuses on liquid state properties and electrochemical characterization of less volatile nonaqueous electrolyte solutions, which may be used as next-generation batteries and electroplating. He is also interested in solid state properties e.g. magnetic and electronic properties of oxides. The studied materials include perovskites and spinels, obtained using structurally-related precursors. Dr. Kitada received a PhD (Dr. Eng.) from Department of Energy and Hydrocarbon Chemistry, Kyoto University in 2012.
Abstract:
Aluminum (Al) metal, known for its low weight and corrosion resistance, finds its applications in structural materials. Smelting of aluminum has been industrialized as the Hall-Héroult process. Electrowinning of aluminum from alumina-dissolved cryolite molten salts is performed at 1000 °C. Instead of the high temperature process, medium and low temperature electrodeposition is fascinating as that can be hired as a new aluminum coating process. Several kinds of medium-low temperature baths for aluminum electroplating are reported using organic solvents (ethers, aromatic hydrocarbons, sulfones) and ionic liquids. Al electroplating is employed for the enhancement of corrosion resistance of magnesium alloys and steel. Additionally, the redox potential of aluminum is low and the theoretical capacity is high. Hence aluminum electroplating baths are captivating as a negative electrode material for the aluminum ion batteries. At electrodeposition temperatures, the usual organic solvents are highly volatile. Since ionic liquids are thermally and chemically stable and also less volatile at room temperature, they are adorable for electroplating baths. On comparing ionic liquids with organic solvents, however, cost of the chemicals required for the bath preparation is high for ionic liquids. Finding a cost effective and safe substitute for aluminum electroplating baths is important. Currently glymes, i.e. glycol ethers, have found to be relatively safe solvents for lithium and magnesium ion batteries, which have boiling points above 150 °C and relatively low volatilities at room temperature.The work reports on room temperature aluminum electrodeposition using aluminum chloride (AlCl3) and diglyme (G2), the first report for aluminum electroplating glyme baths. We also studied the electrochemical properties of AlCl3/glyme solutions using four kinds of glymes. By surprise, electrodeposition was only successful from G2 solutions: only G2 can form electrochemically active Al-Cl-glyme complex cations, which can then undergo easy desolvation of the glymes and subsequent reduction of Al3+.

Toshiaki Kondo
Tokyo Metropolitan University, Japan
Title: Ideally ordered nanohole array obtained by anodizing pretextured metals

Biography:
Toshiaki Kondo works in the field of nano/micro fabrication processing based on electrochemical methods such as anodic oxidation, etching, electroplating and so on. He has proposed several unique fabrication processes of two- and three-dimensionally ordered fine structures and its application to various functional optical devices such as photonic crystals and plasmonic devices. These fabrication processes provide an easy way to obtain various functional devices without applying an electron beam lithography technique
Abstract:
Anodic porous TiO2 has attracted attention due to its wide range of applications such as photonic crystals, solar cells and so on. Performances of the devices depend on its geometrical structures of a nanohole array. Therefore, for improvement of the performances, control of its geometrical structures is important. Until now, improvement of an arrangement of nanoholes in an anodic porous TiO2 by self-ordering anodization process has been achieved. However, it is still difficult to obtain defect-free nanohole array structures in entire sample area. In this presentation, fabrication processes of an anodic porous TiO2 having ideally ordered nanoholes will be presented. An ideally ordered anodic porous TiO2 was obtained by anodizing a pretextured Ti plate. Through a texturing process, an ordered array of concaves was formed on a surface of Ti. In a subsequent anodization, each shallow concave acts as a starting point for generation of a nanohole at initial stage of the anodization and results in an ideally ordered structure. We demonstrated two pretexturing processes. One is a process to directly texture a surface of Ti. The nanopattern was formed by pressing a metal (Ni) mold onto a surface of Ti. The other is a process to texture a surface of Al layered on a Ti. A two-layered sample composed of an Al top layer and a Ti underlying layer was prepared. An ideally ordered array of concaves was formed on the surface of Al by pretexturing process using a Ni mold. Owing to softness of Al, the nanopattern was easily formed. By anodizing the sample, the arrangement of nanoholes was transferred to the surface of Ti substrate, resulting in the ideally ordered anodic porous TiO2 as shown in Fig. 1. These processes are expected to be applied to fabricate various functional devices, which require ideally ordered nanohole structures in TiO2, such as photonic crystals.
Jonathan Coleman
Trinity College Dublin, ireland
Title: All-printed electrochemical thin-film transistors from networks of liquid-exfoliated nanosheets

Biography:
Jonathan Coleman is the Professor of Chemical Physics in the School of Physics and the CRANN and AMBER Research centres, all at Trinity College Dublin. His research involves liquid exfoliation of layered compounds such as graphene, boron nitride and molybdenum disulphide. Exfoliation of these materials gives 2D nanosheets which can easily be processed into thin films or composites from applications from energy storage to sensing to electronics. He has published approximately 250 papers in international journals including Nature and Science, has a h-index of 72 and has been cited ~30000 times. He was recently listed by Thomson Reuters among the world’s top 100 materials scientists of the last decade and was named as the Science Foundation Ireland researcher of the Year in 2011. Prof Coleman has been involved in a number of industry-academic collaborative projects with companies including Hewlett-Packard, Intel, SAB Miller, Nokia-Bell Labs and Thomas Swan..
Abstract:
With the advent of the Internet of Things, the development of printed electronics (PE) is becoming increasingly important, with much research focusing on developing new materials. A number of material sets have been studied, including organics, inorganic nanoparticles and nanotube/nanowire networks. High operating voltages (up to 50V), low mobility (<10 cm2/Vs) and poor current injection are still challenges for organic thin film transistors (OTFTs). Networks of inorganic nanoparticles or nanotubes have demonstrated mobilities and on:off ratios of >10 cm2/Vs and >106 respectively, but may face problems with scalability and integration. These problems have led a number of researchers in the field of 2D materials to attempt to produce printed transistors where the channel material is a network of semiconducting nanosheets. Because of the relatively high mobility of 2D materials, such a network might display mobilities which are competitive or even superior to those achievable with printed organics. In addition, one could envisage all-printed transistors consisting of interconnected networks of semiconducting, conducting and insulating 2D nanosheets. However, switchable nanosheet networks have not been demonstrated. Here, using electrolytic-gating, we demonstrate all-printed, vertically-stacked electrochemical transistors with graphene source, drain and gate electrodes, a transition metal dichalcogenide channel and a BN separator, all formed from nanosheet networks. The BN network contains an ionic liquid within its porous interior that allows electrolytic gating in a solid-like structure. Nanosheet network channels display on-off ratios of up to 600, transconductances exceeding 5 mS and mobilities of >0.1 centimeters squared per volt per second. The on-currents scaled with network thickness and volumetric capacitance as well as the network mobility. In contrast to other devices with comparable mobility, large capacitances, while hindering switching speeds, allow these devices to carry higher currents at relatively low drive voltages.to become trauma- informed that would help this recognition.

Vardan Galstyan
University of Brescia, Italy
Title: Electrochemical surface modification of metal oxides for security applications and healthcare

Biography:
Vardan Galstyan is now researcher at the Sensor Lab, Department of Information Engineering, University of Brescia (Italy). His activity is dedicated to the developments of the synthesis methods using electrochemical approaches for the preparation of nanomaterials and nanocomposites (mainly metal oxides and graphene based materials). He has worked on characterization of the obtained structures for the applications in gas- and biosensors, energy storages, photocatalysis and biomedicine. V. Galstyan is serving as peer reviewer for the different international journals including Journal of Materials Chemistry A, Journal of Materials Chemistry C, ChemComm, ACS Applied Materials & Interfaces, Nanoscale, Physical Chemistry Chemical Physics, Sensors & Actuators: B. Chemical, CrystEngComm, etc.
Abstract:
Statement of the Problem: Chemical sensors have received great attention for the fabrication of small-size and mobile gas sensing devices for the environmental monitoring and clinical diagnostic.1,2 One-dimensional metal oxide nanomaterials are very promising structures for the application in chemical gas sensing.3,4 However, the sensing performance of these materials needs to be improved for the manufacturing of sensing systems with the high sensitivity, selectivity and stability.5 Herein, we report the synthesis of highly ordered titania nanotubes and the modification of their structure and surface for the chemical sensing applications. Methodology & Theoretical Orientation: Well-ordered titania nanotube arrays were prepared by electrochemical anodization. The anodization process was performed in the electrochemical cell with the two-electrode system. Preparation of titania nanotubes by means of electrochemical anodization is anodic formation of nanotubes by oxidation and etching of metallic titanium films. Pt foil was used as a counter electrode. The anodization was carried out by potentiostatic mode at room temperature. This method allows direct growth of the titania nanotubes on different type of substrates and the modification of their surface structure at room temperature. The morphological, structural and elemental analysis of the obtained samples were performed. The sensing properties of the materials were tested towards different, explosive and toxic gases. Findings: Pure and doped nanotubes have been obtained by the modification of anodization parameters. Our studies have shown that the engineering of the band gap and the functionalization of the surface structure of titania enhanced its gas sensing performance. Conclusion & Significance: Investigations have shown that the response and the selectivity of titania nanotubes are improving depending on the modification of their composition and their functionalization. Meanwhile, the developed method is promising for the fabrication of high performance and small size chemical gas sensors.

Chunwen Sun
Chinese Academy of Sciences, China
Title: Recent progress in bifuctional catalysts for rechargeable Li-air batteries

Biography:
Chunwen Sun is a professor at Beijing Institute of Nanoenergy and Nanosystems, Chinese Academy of Sciences.
Abstract:
Among various metal/air batteries, lithium-air batteries possess the highest theoretical gravimetric energy density. However, for the rechargeable Li-air battery, besides the challenges of cost, another issue that has to be addressed in the current technology is the limitations of oxygen reduction reaction (ORR) during discharging process and oxygen evolution reaction (OER) during charging process. The sluggish kinetics of the ORR and OER in Li-air batteries is ascribed to the low efficiency of catalysts. Therefore, the design of a low-cost and stable bifunctional electrocatalyst is a major challenge to the construction of efficient Li-air batteries. Several nanocomposite bifunctional catalysts with low cost have been developed in our lab and studied for lithium-air battery applications. They show enhanced catalytic activity and better long-term durability than the commercial Pt/C catalyst.
- Electrochemical surface science | Batteries and energy sources | Organic and Organometallic Electrochemistry
Session Introduction
Ruiyong Chen
Joint Electrochemistry Lab, KIST, Saarland University,Germany
Title: High voltage aqueous redox flow batteries

Biography:
Dr. Ruiyong Chen received his Ph.D. degree (summa cum laude) in 2011 in physical chemistry from the Saarland University, Saarbrücken, Germany. He is currently a habilitation candidate at the Saarland University and a project leader at the KIST Europe. He has expertise in synthesis and characterization of electrode materials for energy storage systems and energy-efficient electrolysis. He has developed new two lithium intercalation cathode materials with disordered rock-salt structure for improving reversible capacity (up to 400 mAh g-1) for lithium ion batteries. He is currently developing new high voltage aqueous electrolytes for stationary energy storage using redox flow batteries.
Abstract:
Redox flow batteries are promising for large-scale energy storage applications due to their economy and scalability, in comparison with other energy storage systems. In addition, redox flow batteries have unique character of decoupled energy storage and power generation capability. Aqueous redox flow battery systems may offer safe operation, low cost and fast ionic mobility (power generation) in comparison with non-aqueous systems. However, due to the limits in the electrochemical stability window of water, aqueous systems can generally only be operated with low cell voltage (< 1.5 V). Conventional aqueous flow batteries have thus low energy density (< 30 Wh L-1), which is far below that for Li-ion batteries. Non-aqueous electrolytes using organic solvents, although can offer wide electrochemical window, suffer from low ionic conductivity, low solubility for active species and safety concerns. To access high cell voltage in aqueous systems, Chen et al., have recently developed new aqueous ionic liquid electrolytes with widened electrochemical window of about 3 V, using a concentrated aqueous solution of 1-butyl-3-methylimidazolium chloride (BMImCl). Herein, we explore various ionic liquid-based electrolytes and compatible redox couples (soluble species and semi-solid suspension utilizing solid-state cation/anion intercalation reactions) in the attempt to obtain redox flow batteries with high cell voltage and high energy density.
Oliver Clemens
TU Darmstadt, Germany
Title: Intercalation Type Electrode Materials for Fluoride Ion Batteries

Biography:
Oliver Clemens has obtained his PhD in 2012 years from Saarland University, Germany, followed by postdoctoral studies at the University of Birmingham, UK. He is group leader of the Joint Research Laboratory Nanomaterials, TU Darmstadt and KIT, Germany since May 2013. At the beginning of 2017 he became a junior professor (Qualifikationsprofessor) within the Materials Science Department at the TU Darmstadt, Germany within an Emmy Noether Fellowship from the German Research Foundation. He has published more than 36 papers (November 2016) in reputed journals.
Abstract:
Building batteries based on a shuttle of fluoride ions is of interest due to the high stability of fluoride as a charge carrier. Therefore, wide potential windows can be accessible in such battery systems allowing for high energy densities. So far, fluoride ion batteries (FIBs) are mainly fabricated as all solid state batteries using conversion based electrodes. However, conversion reactions are well-known to result in limited battery lifetime due to the large volume changes which arise during the cycling of the battery. In addition, high overpotentials (crystallization, charge transfer) can have a tremendous impact on battery kinetics. To improve battery kinetics, we aim to develop intercalation based fluoride ion batteries with high capacities and high voltage. In this respect, perovskite and perovskite related compounds (e. g. Ruddlesdon-Popper type structures) will be shown to serve as host lattices which can intercalate/deintercalate large amounts of fluoride ions, with theoretical capacities reaching ~ 130 mAh/g and being comparable to lithium ion battery systems. We highlight that such materials can outperform CuF2 as a high voltage cathode in agreement with by DFT based calculations. Latest developments on the identification of suitable anode materials will be described in addition to an overview of current limitations of anion based battery systems. Finallly, we will elucidate structure chemical factors which enable selectivity for the intercalating species for cation (e. g. Li and Na) in comparison to anion based batteries.
Xiangyu Zhao
Nanjing Tech University, China
Title: Rechargeable batteries based on chloride ion transfer

Biography:
Dr. Xiangyu Zhao has his expertise in electrochemical energy storage including electrochemical hydrogen storage and rechargeable batteries such as chloride ion batteries, lithium ion batteries and magnesium batteries. He received his PhD in Materials Science at the Nanjing Tech University in 2010 and then joined the same university. Meanwhile, from 1/2012 to 12/2013, he was awarded by the Guest Scientist Fellowship and worked at Karlsruhe Institute of Technology. He is (co-)author of 9 patent applications and has more than 60 papers published in international journals such as Angew Chem Int Ed, Adv Energy Mater and Energy Environ Sci.
Abstract:
Alternative battery chemistries beyond lithium ion and using abundant electrode materials have been developed. Chloride ion battery (CIB) is a new rechargeable battery based on Cl- anion transfer. This battery shows a variety of electrochemical couples with theoretical energy densities up to 2500 Wh l-1, which is superior to those of conventional lithium ion batteries. Abundant material resources such as Mg, Ca, Na and metal chlorides (e.g., FeCl3, CuCl2 and MgCl2) can be sustainable electrode candidates. The CIB includes a metal chloride/metal electrochemical couple and an ionic liquid electrolyte allowing chloride ion transfer, as reported in the proof-of-principle study of CIB operated at room temperature. The problem is that the metal chloride cathode can react with chloride ions in the electrolyte, leading to the formation of soluble complex anion. This electrode dissolution and the subsequent shuttle would limit the use of metal chloride cathode in the liquid electrolyte system. Metal oxychlorides with higher stability have been proved to be new cathode materials for CIBs. Metal oxychloride/metal systems could also show high theoretical energy densities during the chloride ion transfer. By carbon incorporation in the cathode or optimization of electrolyte composition, more than 70% of the theoretical discharge capacity of single-electron cathode such as FeOCl or VOCl could be delivered. A preliminary study on the multi-electron VOCl2 cathode was also reported in the electrode system using VOCl as cathode and Mg/MgCl2 composite as anode. Besides inorganic electrode materials in rechargeable batteries, organic electrode materials, in particular polymers, have been attracting much attention, due to their advantages of good electrochemical performance, high stability, abundant chemical elements, structural tunability and designing flexibility. Chloride ion doped polymer materilals have been studied and developed as new cathodes for chloride ion batteries. Reversible reversible redox reactions and superior cycling stability were obtained.

Amedeo Capobianco
University of Salerno, italy
Title: Hole transport in oxidized DNA: an integrated approach based on electrochemistry, spectroscopy and quantum chemistry

Biography:
Dr. Amedeo Capobianco earned his PhD in 2008 at University of Salerno, where he now works as an assistant professor. His current research interests include the theoretical treatment of charge transport phenomena in biochemical systems and in organic materials.
Abstract:
Singly ionized DNA exhibits long range hole transport (HT) covering distances up to 200 Å [1]. Hole transport has great biochemical and technological relevance, as it controls the site distribution of the oxidative damage in DNA and makes DNA a potential conduit for nanoelectronics, used both as template and as active component. Different mechanisms for the hole transport have been invoked so far: ion gated transport, thermal activated hopping, and superexchange among others. Independent of the kinetic mechanism, the of hole transfer in oxidized DNA is modulated by the complex mosaic of low-lying electronic states, whose accessibility depends on the effective in-situ hole energies of nucleobases and the electronic couplings between interacting nucleobases. The hole energies for adenine (A) and guanine sites were previously quantified by using voltammetry, the influence of hydrogen bonds on the oxidation potential was also determined. Information for cytosine oxidation was inferred by spectroelectrochemistry experiments. Here we have focused our attention on electronic coupling parameters. The latter have been estimated by the lowering of the oxidation potential observed for DNA oligonucleotides containing an increasing number of stacked homo-purine nucleobases. Voltammetric data were interpreted in terms of the two state model (Figure 1). Experimental results were further confirmed by theoretical computations carried out at the full quantum level for single and double stranded oligonucleotides including sugar-phosphate backbone and solvation. The observed lowering of the oxidation potential for single stranded oligonucleotides containing an increasing number of adjacent adenines amounts to ca 0.3 V for the AA step, in good agreement with theoretical predictions. That result suggests that adenine tracts give rise to delocalized hole domains (Figure 1), greatly facilitating hole transport in DNA [7]. Although stacking interactions are found to be less effective in stabilizing the positive charge on adjacent guanines inside a strand, our results confirm that guanine tracts constitute a very efficient trap for the hole

Teko W. Napporn
University of Poitiers, France
Title: Gold Nanospheres in electrocatalysis: surface interaction with glucose.

Biography:
Dr. Teko W. Napporn is a Researcher of French National Center for Scientific Research (CNRS) and also Adjunct professor at the Institute of Advanced Sciences of Yokohama National University. He is developing nanostructured electrocatalysts for energy conversion (Fuel cells) and storage (batteries and water electrolysis). His research topics deal with the surface structural effect in electrochemistry at electrified Solid-Liquid Interfaces, hybrid biofuel cells. He is also involved in the development of single chamber solid oxide fuel cell.
Abstract:
Gold at nanoscale fascinated scientists who developed important research topics over physics, chemistry, medicine and biotechnology. The last three decades, various innovative investigations emerged for determining the role of size, the morphology on the unexpected properties observed for gold at nanoscale. In electrocatalysis, gold nanoparticles (AuNPs) were often used to understand the intrinsic relationship between their size, morphology and activity towards two main reactions: (i) the electrooxidation of organic molecules that have a great interest as fuel in fuel cell applications, and (ii) the oxygen reduction reaction (ORR). Electrocatalysis investigations on support-less gold nanorods have shown that it is a challenge to study the intrinsic properties of their surface through their interaction with a reactive molecule. Our recent results on gold nanospheres revealed that the size of these nanomaterials plays a key role in their electrochemical response. Therefore spherical gold nanoparticles (AuNSs) with a mean diameter from 4 to 15 nm were successfully synthesized. UV-visible spectroscopy, Transmission electron microscopy, and Under-Potential Deposition of lead (UPD) were used for determining their morphology, size and approaching their surface crystallographic structure. UPD of lead reveals that their crystallographic facets are affected by their size and the growth process. In alkaline medium, the oxidation of glucose was used to evaluate their electroactivity. As results, small AuNSs exhibited drastic increase of catalytic activity (fig. 1). This feature might result in the high specific area and reactivity of the surface electron induced by their small size. The study of the reaction mechanism was investigated by in situ Fourier transform infrared reflectance spectroscopy. Gluconolactone and gluconate were identified respectively as the intermediate and the final reaction product of the glucose electrooxidation.

Milko E. van der Boom
The Weizmann Institute of Science
Title: Electron Transfer in Coordination-based Molecular Assemblies

Biography:
Milko van der Boom completed his B.Sc. in Chemical Engineering at the Amsterdam University of Applied Sciences and his M.Sc. degree in Inorganic Chemistry at the University of Amsterdam. In 1994, he enrolled as a doctoral student at the Weizmann Institute of Science, where he studied Organometallic Chemistry; he was awarded his Ph.D. degree with distinction in 1999. After three years of postdoctoral work at Northwestern University, where he studied the formation of functional organic films, he returned as a faculty member to the Weizmann Institute’s Department of Organic Chemistry. His interdisciplinary materials chemistry research focuses on metalloorganic-oriented synthetic and mechanistic studies. Prof. van der Boom's prizes and honors include an Alon Fellowship from the Israel Council for Higher Education, the Henri Gutwirth Prize from the Technion, and the Israel Chemistry Society's Prize for Excellent Young Chemists. He is the first incumbent of the Bruce A. Pearlman Professional Chair.
Abstract:
Directional electron-transfer events are the basis of many technologically important systems and biological processes. In this study, we demonstrate how the distance over which electron transfer occurs through organic materials can be controlled and extended. Coating of conductive surfaces with nanoscale layers of redox-active metal complexes allows the electrochemical addressing of additional but distant layers that are otherwise electrochemically silent. We also show that our composite materials can pass electrons selectively in directions that are determined by the positioning of redox-active metal complexes and the distances between them. These electron-transfer processes can be made dominantly uni- or bidirectional. Our design strategy involves 1) a set of isostructurally well-defined metal complexes with different electron affinities, 2) a scalable metal-organic spacer, and 3) a versatile assembly approach that allows systematic variation of material composition, structure, and electron transfer properties. We control the electrochemical communication between interfaces by the deposition sequence of the components and the length of the spacer, and therefore we are able to program the bulk properties of the assemblies.

Michal Lahav
The Weizmann Institute of Science, Israel
Title: Metalloorganic Assemblies as Electrochromic Materials: Switching Stability, Coloration Efficiencies and Devices

Biography:
Michal Lahav completed her BSc and PhD studies in Chemistry (cumma sum laude) in 2001 at the Hebrew University of Jerusalem. She was a postdoctoral researcher at the Weizmann Institute of Science for two years before she moved to Harvard University, where she studied nanochemistry. After two years of postdoctoral work in the United States, she returned to Israel and started to work as a scientific advisor at the Weizmann Institute of Science. She was appointed as an Associate Staff Scientist in the Department of Organic Chemistry in 2011. Her interdisciplinary materials chemistry research is related to the self-assembly of metallo-organic materials for energy storage and for electrochromics whose products are now being patented. Her work is related to fundamental understanding of the formation and electronic properties of these metallo-supramolecular architectures. Dr. Michal Lahav’s prizes and honors include the Dr. Maxine Singer Prize for Outstanding Staff Scientists, the Baruch Zinger Award for Academic Excellency, the IVS Excellence Award for Surface Science Expertise and the Schmidt Prize.
Abstract:
Stepwise deposition from solution, combined with metal-ligand coordination, has served as a powerful tool for generating functional architectures on surfaces. Such systems might find many applications in molecular electronics, sensor, and solar cells. More significantly, owing to their interesting electrochromic (EC) behavior, redox-active metallo-organic assemblies are promising candidates for use in smart windows. In this study, we used a dip-coating process to generate EC molecular assemblies (MA) from metal polypyridyl complexes cross-linked with a palladium salt. These complexes are considered ideal chromophores for fabricating EC materials, due to their excellent stability and light absorption that significantly depends on their oxidation state. Varying the number of pyridine moieties was used to control (i) the materials’ stability, (ii) color, (iii) redox-chemistry, and (iv) the film growth (i.e., linear vs. exponential). Our observations also demonstrated that minor structural differences (i.e., the pyridine-bipyridine bond order, X) at the molecular level become apparent in stability and EC properties, (Figure 1). The MAs exhibit very high coloration efficiencies (CEs) and are extremely stable. Furthermore, we demonstrate solid-state devices.
Marie-Christine Record
Aix-Marseille University, France
Title: Electrochemical Atomic Layer Deposition (EC-ALD) of Semiconducting Materials

Biography:
Marie-Christine Record has completed her PhD in 1992 from Montpellier University (France) and postdoctoral studies from Ecole Centrale Paris (France). She was an associate professor at Montpellier University from 1996 to 2004 and from 2004, she is full professor at the Aix-Marseille University (France) where she works on the elaboration of thin films materials for energy. She has published more than 80 papers in reputed journals and has been serving as an editorial board member of repute.
Abstract:
Electrochemical deposition may provide an alternative process to physical deposition methods for peparing semiconducting thin films. Indeed by contrast to other methods, electrochemical deposition is a low cost, room temperature production technique, which works without vacuum atmosphere and allows one to cover substrates with complex shapes. The shortcoming of electrochemical deposition is limited control on the size, stoichiometry and crystallinity of deposits. The electrochemical atomic layer deposition (EC-ALD) method was put forward in 1991 by Gregory and Stickney. This method is based on the alternate underpotential deposition (UPD) of atomic layers of the elements to make up a compound, combining advantageously the technique of electrochemical deposition and that of atomic layer deposition (ALD). Underpotential deposition is a surface-limited electrochemical phenomenon, which makes the deposition generally limited to an atomic layer. In every cycle one monolayer of the compound is obtained, and the thickness of the deposit will only depend on the number of cycles. Each cycle consists in a series of individual steps, and each step can be optimized independently, resulting in well-controlled deposits. In the first part of this talk, I will describe the EC-ALD method and I will expose the advantages of this method compared to those of physical ones of ALD type and compared to those of other electrochemical methods. Then, I will detail the EC-ALD experiment platform designed and constructed in our group. Finally, I will present some results we got with this equipment for the electrodeposition of CoSb3 and Sb2Se3 compounds.
Mitsuharu Tabuchi
National Institute of Advanced Industrial Science and Technology (AIST), Japan
Title: Synthesis and characterization of Ni- and Ti substituted Li2MnO3 positive electrode material for lithium-ion battery

Biography:
Mitsuharu Tabuchi is an inorganic synthetic chemist using wet-chemical process for oxide-based positive electrode material of lithium-ion battery. His synthetic experiences above 15 years were constructed mainly to get homogeneous LiFeO2-Li2MnO3 solid solution. The constructed synthetic route is also effective for preparing LiNi1/2Mn1/2O2-Li2MnO3 one. He has also characterization experience using 57Fe Mössbauer spectroscopy and X-ray Rietveld analysis to know charge compensation mechanism for LiFeO2-Li2MnO3 system. He found also Fe and Ni substituted Li2MnO3 and it has high-potential for practical use, because it can be prepared largely by chemical company (Tanaka Chemical Corporation) and then is electrochemically tested by battery manufacturer (NEC Corporation) as power-source of electronic vehicle (EV).
Abstract:
Li-excess positive electrode material (LiMO2-Li2MnO3 solid solution, M=Ni1/2Mn1/2 or Ni1/3Mn1/3Co1/3) is a good candidate as its high-capacity (250 mAh/g) and acceptable discharge voltage above 3.5 V for high energy-density lithium-ion battery to EV and PHEV application. Especially LiNi1/2Mn1/2O2-Li2MnO3 solid solution (Li1+x(NiyMn1-y)1-xO2, 0

Yu-Wu Zhong
Institute of Chemistry, Chinese Academy of Sciences, china
Title: Electro-Active Ruthenium-Amine Conjugated Organometallic Materials

Biography:
Yu-Wu Zhong obtained his Ph.D. in July 2004 from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences (CAS) under the supervision of Prof. Guo-Qiang Lin. From September 2004 to September 2006, he has been a postdoctoral researcher at the University of Tokyo with Prof. Eiichi Nakamura. From October 2006 to September 2009, he worked with Prof Hector D. Abruna at Cornell University as a postdoctoral researcher. In October 2009, he was awarded by the “100 talent” program of CAS and joined Key Laboratory of Photochemistry of the Institute of Chemistry, CAS, to start his independent career. His research interests focus on electro-active and photofunctional organic materials and transition-metal complexes. He has published more that 110 peer-reviewed papers to date. He is currently an associate editor of RSC Advances and editorial board member of Scientific Reports and Science China Chemistry.
Abstract:
Electro-active and electrochromic materials have received a wide range of applications. The incorporation of a metal ion gives rise to new functions that are not present in common organic materials. However, the applications of electro-active organometallic materials are often hampered by their high redox potentials and difficulty in film formation. Recently, we have been interested in the design and construction of electro-active systems with cyclometalated ruthenium and triarylamine as the charge-bearing sites. These materials show strong electronic coupling and multiple reversible redox processes in low positive potential window. After modified with vinyl or triphenyl units, these can be smoothly deposited onto ITO electrode surfaces by in situ electropolymerization. The obtained films exhibit multistate NIR electrochromism with long retention time, good contrast ratio, and low switching potential. The response is about a few seconds. Depending on the number of redox sites, 3 - 5 step redox switching were realized. These films have been used for the demonstrations of flip-flop or flip-flap-flop memory with electrochemical potentials as input signals and absorbance at different wavelengths as output signals. In addition to electropolymerized films, self-assembled monolayer films of these complexes were obtained and they are useful in molecular-scale NIR electrochromism and electrochemically-gated single molecular conductance

Yan Jiao
The University of Adelaide, Australia
Title: Evaluation of doped graphene materials for electrochemical hydrogen evolution reaction and oxygen reduction reaction

Biography:
Dr Yan Jiao's research interests involve discovering the origin of electrocatalytic activity possessed by carbon-based materials by computational chemistry. She also aims to design novel carbon-based catalysts for clean energy conversion reactions, including hydrogen evolution reaction, oxygen reduction reaction and carbon dioxide reduction reaction. She obtained her PhD in Chemical Engineering from the University of Queensland, and is currently working as a postdoctoral researcher in the University of Adelaide (UoA). She is the receiver of several awards, including Women's Research Excellence Award by UoA. She has received over 4400 citations and h-index is 19.
Abstract:
The dwindling supply of fossil fuels urge us to explore alternative power sources to drive our highly automotive society. Under this background, establish reliable clean and sustainable energy supplies are of great importance, and using electrochemical method to realize energy conversions hold a great promise. Among these reactions, hydrogen evolution reaction (HER) and oxygen reduction reaction (ORR) are the most studied, due to their fundamentality in electrocatalysis and their role in hydrogen production and fuel cells, respectively. Effective candidates for these two reactions are often based on noble metals, while carbon-based metal-free electrocatalysts generally demonstrate poorer activity. Here we report evaluation of a series of heteroatom-doped graphene materials as efficient HER and ORR electrocatalysts by density functional theory calculations, with the input of spectroscopic characterizations and electrochemical measurements. Results of theoretical computations are shown to be in good agreement with experimental observations regarding the intrinsic electrocatalytic activity and the reaction mechanisms for these two reactions. As a result, we establish volcano shaped activity trends for HER and ORR on graphene-based materials, and explore their reactivity origin to guide the design of more efficient electrocatalysts. We predict that by rationally modifying particular experimentally achievable physicochemical characteristics, a practically realizable graphene-based material will have the potential to exceed the performance of the metal-based benchmarks for these two reactions.
Sylvio Indris
Karlsruhe Institute of Technology, Institute for Applied Materials – Energy Storage Systems, Germany
Title: Electrodes and Solid Electrolytes for Li-Ion Batteries: Local Structures, Li-Ion Mobility, and Lithiation Mechanisms

Biography:
Sylvio Indris is investigating materials that can be used as electrodes and electrolytes in Li-ion batteries. These materials are prepared by solvothermal/hydrothermal methods, sol-gel techniques, and solid-state reactions. The changes in these materials during operation in these batteries are investigated by diffraction techniques as well as X-ray absorption spectroscopy, solid-state NMR, and Mössbauer spectroscopy. Finally, the results of these investigations are used to optimize the performance of the complete Li-ion battery systems.
Abstract:
Li-ion batteries are used extensively in mobile electronic devices and in electric vehicles due to their high energy and power densities. The performance of these storage systems strongly depends on the materials used inside these batteries as electrodes and electrolytes. In order to improve these batteries, it is important to understand the fundamental diffusion mechanisms and also the fundamental electrochemical reaction mechanisms that occur during charging and discharging of the batteries, i.e. during lithiation and delithiation of the electrodes, and that are responsible for function and degradation of these systems. The changes that occur in the electrode materials during electrochemical cycling are investigated by in situ techniques including diffraction as well as spectroscopic methods [1-3]. These measurements are performed on olivine materials LiMPO4 (M = Fe, Mn, Co) and high-voltage spinel materials LiM’M’’O4 (M’, M’’ = Mn, Co, Ni, Fe) and they reveal the details of the lithiation/delithiation mechanisms and how these are related to the performance of these materials in the batteries. Solid electrolytes might offer an enhanced safety for these systems. Different oxidic, sulfidic, and polymer systems are investigated with respect to Li+ ion mobility by 7Li NMR relaxometry and field-gradient NMR spectroscopy [4-6]. From this, microscopic diffusion parameters such as the average Li ion jump rate t-1 and the activation barrier EA for single Li ion jumps can be extracted, but also the macroscopic transport of the Li ions can be observed. The results of these measurements are compared to those obtained by impedance spectroscopy.
- Electrochemical surface science | Batteries and energy sources | Organic and Organometallic Electrochemistry | Corrosion Science and Technology | Physical and Analytical Electrochemistry
Location: Berlin, Germany
Session Introduction
Junhua Dong
Professor
Title: Study on the galvanic corrosion of ferrite-pearlite steel and its control under an acid environment

Biography:
Dong Junhua is a professor working in Institute of Metal Research (IMR), Chinese Academy of Sciences (CAS). He has his expertise in electrochemical corrosion principle of metals in study and teaching. His primary research aim is to solve various industrial and natural environmental corrosion problems. His research interests has been focused on: the evaluation of atmospheric corrosion evolution of low alloy steels; the study of developing cost effective weathering steel and marine corrosion resistant steel; the design of waste containers for the underground geological disposal of high level nuclear wastes in China; the study of marine corrosion in tidal zone and deep sea; the evaluation of concrete corrosion; the study of magnesium corrosion and protection by alloying and coating technique; the evaluation of corrosion inhibitors; electrochemical corrosion monitoring for various corrosion environments. He has taught the course of electrochemical corrosion principle of metals to the graduate students for 14 years in his organization.
Abstract:
Statement of the Problem: Low carbon steel with the microstructures of ferrite and pearlite phases is the most widely used engineering structure material, in which pearlite with a typical lamellar structure consisting of alternating layers of ferrite and cementite (Fe3C) plays an important role in the mechanical properties of steel. However, as the electrically contact of Fe3C as a cathode site and ferrite site as anode in electrolyte can cause the galvanic corrosion, the corrosion resistance of steels will be rapidly deteriorated. The purpose of this study is to describe the sustained effect of the remaining cementite on enhancing the corrosion deterioration and the effect of several added alloy elements on retarding the corrosion of low carbon steel in an acid solution. Methodology & Theoretical Orientation: Immersion test, X-ray diffraction, micromorphology observation, potentio-dynamic polarization, open circuit potential monitoring and electrochemical impedance spectroscopy was utilized in this study. Findings: The results show that, with extending the immersion time, the preferential dissolution of the exposed ferrite resulted in a lot of cementite accumulation on the steel surface, which enhanced the galvanic effect and hence accelerated the corrosion. The promoted anodic dissolution of ferrite phases was controlled by charge transfer process, while the hydrogen evolution reduction occurred on cementite was controlled by the diffusion process. For 16MnCu steel, elemental Cu precipitated as the nano-sized particles on the steel surface, which improved the corrosion resistance of the steel by weakening the galvanic effect between the ferrite and cementite sites. Moreover, trace of alloy elements of Sn or Sn-Mo could improve the corrosion resistance of AH32 steel, which is also ascribed to that Sn or Sn-Mo could retard the galvanic corrosion between the ferrite and cementite phases. Sn and Mo should exist as the metallic state and were uniformly distributed on the steel surface.


Biography:
Dr. Ruiyong Chen received his Ph.D. degree (summa cum laude) in 2011 in physical chemistry from the Saarland University, Saarbrücken, Germany. He is currently a habilitation candidate at the Saarland University and a project leader at the KIST Europe. He has expertise in synthesis and characterization of electrode materials for energy storage systems and energy-efficient electrolysis. He has developed new two lithium intercalation cathode materials with disordered rock-salt structure for improving reversible capacity (up to 400 mAh g-1) for lithium ion batteries. He is currently developing new high voltage aqueous electrolytes for stationary energy storage using redox flow batteries.
Abstract:
Redox flow batteries are promising for large-scale energy storage applications due to their economy and scalability, in comparison with other energy storage systems. In addition, redox flow batteries have unique character of decoupled energy storage and power generation capability. Aqueous redox flow battery systems may offer safe operation, low cost and fast ionic mobility (power generation) in comparison with non-aqueous systems. However, due to the limits in the electrochemical stability window of water, aqueous systems can generally only be operated with low cell voltage (< 1.5 V). Conventional aqueous flow batteries have thus low energy density (< 30 Wh L-1), which is far below that for Li-ion batteries. Non-aqueous electrolytes using organic solvents, although can offer wide electrochemical window, suffer from low ionic conductivity, low solubility for active species and safety concerns. To access high cell voltage in aqueous systems, Chen et al., have recently developed new aqueous ionic liquid electrolytes with widened electrochemical window of about 3 V, using a concentrated aqueous solution of 1-butyl-3-methylimidazolium chloride (BMImCl). Herein, we explore various ionic liquid-based electrolytes and compatible redox couples (soluble species and semi-solid suspension utilizing solid-state cation/anion intercalation reactions) in the attempt to obtain redox flow batteries with high cell voltage and high energy density.

Biography:
Oliver Clemens has obtained his PhD in 2012 years from Saarland University, Germany, followed by postdoctoral studies at the University of Birmingham, UK. He is group leader of the Joint Research Laboratory Nanomaterials, TU Darmstadt and KIT, Germany since May 2013. At the beginning of 2017 he became a junior professor (Qualifikationsprofessor) within the Materials Science Department at the TU Darmstadt, Germany within an Emmy Noether Fellowship from the German Research Foundation. He has published more than 36 papers (November 2016) in reputed journals
Abstract:
Building batteries based on a shuttle of fluoride ions is of interest due to the high stability of fluoride as a charge carrier. Therefore, wide potential windows can be accessible in such battery systems allowing for high energy densities. So far, fluoride ion batteries (FIBs) are mainly fabricated as all solid state batteries using conversion based electrodes. However, conversion reactions are well-known to result in limited battery lifetime due to the large volume changes which arise during the cycling of the battery. In addition, high overpotentials (crystallization, charge transfer) can have a tremendous impact on battery kinetics.To improve battery kinetics, we aim to develop intercalation based fluoride ion batteries with high capacities and high voltage. In this respect, perovskite and perovskite related compounds (e. g. Ruddlesdon-Popper type structures) will be shown to serve as host lattices which can intercalate/deintercalate large amounts of fluoride ions, with theoretical capacities reaching ~ 130 mAh/g and being comparable to lithium ion battery systems. We highlight that such materials can outperform CuF2 as a high voltage cathode in agreement with by DFT based calculations. Latest developments on the identification of suitable anode materials will be described in addition to an overview of current limitations of anion based battery systems. Finallly, we will elucidate structure chemical factors which enable selectivity for the intercalating species for cation (e. g. Li and Na) in comparison to anion based batteries.

Biography:
Dr. Xiangyu Zhao has his expertise in electrochemical energy storage including electrochemical hydrogen storage and rechargeable batteries such as chloride ion batteries, lithium ion batteries and magnesium batteries. He received his PhD in Materials Science at the Nanjing Tech University in 2010 and then joined the same university. Meanwhile, from 1/2012 to 12/2013, he was awarded by the Guest Scientist Fellowship and worked at Karlsruhe Institute of Technology. He is (co-)author of 9 patent applications and has more than 60 papers published in international journals such as Angew Chem Int Ed, Adv Energy Mater and Energy Environ Sci.
Abstract:
Alternative battery chemistries beyond lithium ion and using abundant electrode materials have been developed. Chloride ion battery (CIB) is a new rechargeable battery based on Cl- anion transfer. This battery shows a variety of electrochemical couples with theoretical energy densities up to 2500 Wh l-1, which is superior to those of conventional lithium ion batteries. Abundant material resources such as Mg, Ca, Na and metal chlorides (e.g., FeCl3, CuCl2 and MgCl2) can be sustainable electrode candidates. The CIB includes a metal chloride/metal electrochemical couple and an ionic liquid electrolyte allowing chloride ion transfer, as reported in the proof-of-principle study of CIB operated at room temperature. The problem is that the metal chloride cathode can react with chloride ions in the electrolyte, leading to the formation of soluble complex anion. This electrode dissolution and the subsequent shuttle would limit the use of metal chloride cathode in the liquid electrolyte system. Metal oxychlorides with higher stability have been proved to be new cathode materials for CIBs. Metal oxychloride/metal systems could also show high theoretical energy densities during the chloride ion transfer. By carbon incorporation in the cathode or optimization of electrolyte composition, more than 70% of the theoretical discharge capacity of single-electron cathode such as FeOCl or VOCl could be delivered. A preliminary study on the multi-electron VOCl2 cathode was also reported in the electrode system using VOCl as cathode and Mg/MgCl2 composite as anode. Besides inorganic electrode materials in rechargeable batteries, organic electrode materials, in particular polymers, have been attracting much attention, due to their advantages of good electrochemical performance, high stability, abundant chemical elements, structural tunability and designing flexibility. Chloride ion doped polymer materilals have been studied and developed as new cathodes for chloride ion batteries. Reversible reversible redox reactions and superior cycling stability were obtained

Biography:
Dr. Teko W. Napporn is a Researcher of French National Center for Scientific Research (CNRS) and also Adjunct professor at the Institute of Advanced Sciences of Yokohama National University. He is developing nanostructured electrocatalysts for energy conversion (Fuel cells) and storage (batteries and water electrolysis). His research topics deal with the surface structural effect in electrochemistry at electrified Solid-Liquid Interfaces, hybrid biofuel cells. He is also involved in the development of single chamber solid oxide fuel cell.
Abstract:
Gold at nanoscale fascinated scientists who developed important research topics over physics, chemistry, medicine and biotechnology. The last three decades, various innovative investigations emerged for determining the role of size, the morphology on the unexpected properties observed for gold at nanoscale. In electrocatalysis, gold nanoparticles (AuNPs) were often used to understand the intrinsic relationship between their size, morphology and activity towards two main reactions: (i) the electrooxidation of organic molecules that have a great interest as fuel in fuel cell applications, and (ii) the oxygen reduction reaction (ORR). Electrocatalysis investigations on support-less gold nanorods have shown that it is a challenge to study the intrinsic properties of their surface through their interaction with a reactive molecule. Our recent results on gold nanospheres revealed that the size of these nanomaterials plays a key role in their electrochemical response. Therefore spherical gold nanoparticles (AuNSs) with a mean diameter from 4 to 15 nm were successfully synthesized. UV-visible spectroscopy, Transmission electron microscopy, and Under-Potential Deposition of lead (UPD) were used for determining their morphology, size and approaching their surface crystallographic structure. UPD of lead reveals that their crystallographic facets are affected by their size and the growth process. In alkaline medium, the oxidation of glucose was used to evaluate their electroactivity. As results, small AuNSs exhibited drastic increase of catalytic activity (fig. 1). This feature might result in the high specific area and reactivity of the surface electron induced by their small size. The study of the reaction mechanism was investigated by in situ Fourier transform infrared reflectance spectroscopy. Gluconolactone and gluconate were identified respectively as the intermediate and the final reaction product of the glucose electrooxidation.

Biography:
Benoît Ter-Ovanessian has been named associate professor in the Material Science and Engineering Department at INSA de Lyon in France in 2013. He has joined the Corrosion Science and Surface Engineering group of the MATEIS laboratory and integrated the research project of the CNRS International Associated Laboratory, ELyT Lab, which promotes collaborations between Lyon University (Fr) and Tohoku University (Jp). His research activities are mainly focused on the understanding of the passivation features, the interactions between mechanical loading and interfacial reactivity; and consequently the managing of the reliability of metallic components.
Abstract:
Semi-conductive properties of passive film represent discriminant parameters in many applications in photo-electrochemistry such as photoanodes or photoelectrochemical cells, or in electrochemistry such as sensors, bio-electrochemical cells and corrosion. For this reason, the accurate determination of these properties is of great interest. Considering passive films as thin highly disordered semiconductor, the electronic properties of passive films are generally investigated by photo-electrochemistry or differential capacitance measurement of the Mott-Schottky (MS) theory. In the MS approach, electrochemical impedance spectroscopy (EIS) is a widely used technique to determine the capacitance of the passive film. However, different limitations are known to alter the final results. For example, the electronic properties obtained by conventional MS experiments are known to be frequency dependent. Furthermore, as Constant-Phase-Element (CPE) behavior is commonly observed on the impedance diagrams recorded with passive materials, the method to extract the effective capacitance from the high frequency time-constant dispersions has to be carefully chosen. In the present work, the recent advances in experimental procedure and interpretations of EIS spectra during MS experiments, which allow to accurately determine the electronics properties of passive film, are described for passive films grown on pure Chromium and Nickel-Chromium alloys. The relevance of multi-frequency MS experiments that limits the frequency dependency of the capacitance measurements is firstly discussed. Secondly, the pertinence of the power-law model or the Cole-Cole representation of the complex capacitance to assess the passive film capacitance from EIS diagrams during MS experiments is then debated for different systems

Biography:
Elise Deunf received her PhD in physical and analytical chemistry in 2010 under Dr Christian Amatore from the Ecole Normale Supérieure (Paris). She then joined the Lawrence Berkeley National Laboratory and University of California Berkeley as a postdoctoral fellow to develop new organic hydrogen carriers for PEM-fuel cells in collaboration with GE Global research. In 2013, she moved back to France to work with Prof. Philippe Poizot at the Institute of Materials Jean Rouxel (Nantes). Her current research focuses on molecular electrochemistry and the development of organic electrode materials for alternative and low-polluting batteries.
Abstract:
Routine access to power sources is an essential factor for developing our technology-oriented society and ensuring a better quality of life. In this context, while the implementation of renewable energy sources is in progress, electrical energy storage (EES) systems are set to play a central and potentially critical role in the next-generation energy infrastructure. Faced with a worldwide demand for powering electrified vehicles and electronic devices of all kinds, accelerated progress and innovation in the development of new and potentially “greener” electrochemical storage solutions are thus imperative. Based on the tailoring of naturally abundant chemical elements (C, H, N, O, S, in particular), organic chemistry provides great opportunities for finding innovative electrode materials able to operate in aqueous or nonaqueous electrolytes.1 Along this line, significant progress has been achieved these last ten years on redox-active organic compounds, bringing them to the attention of the energy storage community.
Indeed, organic chemistry provides great opportunities for discovering innovative electrode materials able to operate both in aqueous and nonaqueous electrolytes. Additionally, it must be pointed out that two types of electrochemical insertion mechanisms can be used in practice with either reversible uptake/release of cations or anions (figure 1a, 1b respectively). In this communication, we will present recent advances on organic host materials and will particularly report on small organic compounds based on aromatic core structures that are functionalized by redox-active groups such as enolate and amine, and which able to reversibly host cations or anions by electrochemical reaction

Biography:
Huriye Ä°cil was borned in Larnaca, Cyprus, in 1960. She received her Ph.D in Organic Chemistry from Ege University of Turkey, in 1993. In 1993, 1995 and 2002 she was appointed as Assist. Professor, Assoc. Professor and full Professor, respectively at Eastern Mediterranean Univ. (North-Cyprus). She directed many research projects funded by NATO, DFG, DAAD, CNRS, TUBITAK and DPT. She held Visiting Scientists positions at Max Planck Institute (Mulheim), Rochester Univ., and East Anglia Univ. She has 32 published articles in reputed journals. She graduated 33 MSc and 9 PhD students. Her research interests focus on design and synthesis of novel organic materials with remarkable electrical and optical properties.
Abstract:
Development of novel materials with optimal structural, thermal, photochemical and electrochemical properties plays an important role in the researches for a sustainable energy future. Perylene dyes are particularly important compounds due to their remarkable absorption and emission in the spectral region from visible to near infrared (NIR), photochemical and electrochemical properties, and thermal, chemical and photochemical stabilities. They are indeed very promising electron acceptors for efficient organic photovoltaics (OPV). Undoubtedly, photovoltaic applicability of perylene dyes mainly based electrochemical properties. In general, perylene dyes show two different reversible and electrochemically stable reduction peaks in solution state. The first reduction involves the conversion of the neutral compound to the relevant radical anion and the second one accounts for the further reduction to dianion. The separation between the potentials at which the two charge transfers take place allows one to discard the occurrence of any redox reaction between the dianion and the neutral compound. Interestingly the electrochemical characteristics of perylene dyes are completely different in solutions and solid state. They show only one broad and reversible reduction peak in solid state. Further, the analysis of the voltammetric signals suggested that two anodic and cathodic peak systems were actually overlapped. Planarity of the structure, introducing electron donor and acceptor substituents at the imide or bay positions or different intermolecular interactions caused different electrochemical properties. In this study, a detailed comparative study on the electrochemical properties of N-substituted or bay substituted perylene diimides, donor-acceptor-donor structured perylene diimides, self-assembled microstructure perylene diimides and perylene polymers, has been carried out. For comparison, the HOMO-LUMO energy differences for the selected perylene dyes were determined using both electrochemical and optical methods. Notably, the HOMO energy values obtained through the electrochemical and optical band gaps are quite different.

Biography:
Yu-Wu Zhong obtained his Ph.D. in July 2004 from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences (CAS) under the supervision of Prof. Guo-Qiang Lin. From September 2004 to September 2006, he has been a postdoctoral researcher at the University of Tokyo with Prof. Eiichi Nakamura. From October 2006 to September 2009, he worked with Prof Hector D. Abruna at Cornell University as a postdoctoral researcher. In October 2009, he was awarded by the “100 talent” program of CAS and joined Key Laboratory of Photochemistry of the Institute of Chemistry, CAS, to start his independent career. His research interests focus on electro-active and photofunctional organic materials and transition-metal complexes. He has published more that 110 peer-reviewed papers to date. He is currently an associate editor of RSC Advances and editorial board member of Scientific Reports and Science China Chemistry.
Abstract:
Electro-active and electrochromic materials have received a wide range of applications. The incorporation of a metal ion gives rise to new functions that are not present in common organic materials. However, the applications of electro-active organometallic materials are often hampered by their high redox potentials and difficulty in film formation. Recently, we have been interested in the design and construction of electro-active systems with cyclometalated ruthenium and triarylamine as the charge-bearing sites. These materials show strong electronic coupling and multiple reversible redox processes in low positive potential window. After modified with vinyl or triphenyl units, these can be smoothly deposited onto ITO electrode surfaces by in situ electropolymerization. The obtained films exhibit multistate NIR electrochromism with long retention time, good contrast ratio, and low switching potential. The response is about a few seconds. Depending on the number of redox sites, 3 - 5 step redox switching were realized. These films have been used for the demonstrations of flip-flop or flip-flap-flop memory with electrochemical potentials as input signals and absorbance at different wavelengths as output signals. In addition to electropolymerized films, self-assembled monolayer films of these complexes were obtained and they are useful in molecular-scale NIR electrochromism and electrochemically-gated single molecular conductance.

































